In the modern era of orthopedic surgery, Cannulated Screws have transitioned from specialized tools to fundamental components of fracture fixation and reconstructive procedures. As a leading Cannulated Screws Manufacturer, we observe that the global market is witnessing an unprecedented surge. The primary driver is the shift toward Minimally Invasive Surgery (MIS). Cannulated screws allow surgeons to place a guide wire first under fluoroscopy, ensuring precision before the final screw is inserted. This reduces surgical trauma, minimizes blood loss, and accelerates patient recovery.
Geographically, North America and Europe remain the largest consumers due to advanced healthcare infrastructure and an aging population prone to hip fractures and osteoporotic conditions. However, the Asia-Pacific region, led by China and India, is the fastest-growing market. This growth is fueled by increasing healthcare expenditure, a rise in sports-related injuries, and the expansion of specialized orthopedic hospitals requiring high-volume, high-quality implants from reliable manufacturers.
The evolution of cannulated screw manufacturing is deeply intertwined with material science. While Stainless Steel 316L remains a cost-effective choice for many regions, Titanium Alloy (Ti-6Al-4V ELI) has become the gold standard. Titanium offers superior biocompatibility, a lower modulus of elasticity (closer to human bone), and MRI compatibility, which is crucial for post-operative monitoring.
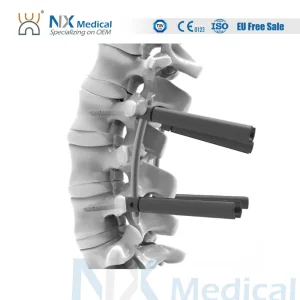
Our advanced 5.5mm system (view product here) represents the pinnacle of AI-guided manufacturing. Designed for cement augmentation, these screws provide unparalleled stability in osteoporotic spinal fixations.
Recent trends also include the development of PEEK (Polyetheretherketone) interference screws and bioabsorbable materials for sports medicine, particularly ACL and PCL reconstructions. As manufacturers, we are integrating smart machining processes to create specialized thread designs—such as self-tapping and self-drilling tips—that reduce the torque required for insertion and prevent bone necrosis.
Furthermore, the integration of 3D printing and digital twin technology in the manufacturing process allows for custom-designed cannulated screws for complex reconstructive surgeries. This level of customization is becoming a key competitive edge for top-tier manufacturers in the global orthopedic supply chain.
Procurement departments in international hospitals and large-scale medical distributors prioritize three main factors: Regulatory Compliance (CE/ISO/FDA), Supply Chain Reliability, and Technical Versatility. In regions like Latin America and the Middle East, there is a massive demand for trauma-focused cannulated screws (4.0mm, 6.5mm, and 7.3mm sizes) to address the high incidence of road traffic accidents and geriatric femoral neck fractures.
Localized application scenarios vary significantly:
As a Cannulated Screws Manufacturer, we provide comprehensive surgical kits alongside our implants, ensuring that local surgeons have the exact instruments—guide wires, cannulated drills, and depth gauges—needed for successful outcomes.
Operating from Guangdong, the heart of China’s high-tech manufacturing hub, Guangdong Marin-one Medical Devices Co., Ltd. combines 18+ years of R&D with cost-efficiencies that Western manufacturers cannot match. Our Titanium Bone Screws (view product details) undergo rigorous fatigue testing and surface passivation to ensure longevity and safety.
Our factory utilizes Swiss-type CNC lathes and automated inspection systems, ensuring that every screw—from the hollow core to the micro-thread—meets micron-level tolerances. This precision is why global buyers choose us for OEM and ODM projects.

Beyond the technical specifications, our advantage lies in our Integrated Ecosystem. We don't just sell a screw; we provide a full spinal, trauma, and joint system. With a production philosophy of "Innovation First," we are constantly updating our designs based on feedback from international orthopedic surgeons, ensuring our products are always at the forefront of clinical efficacy.
Guangdong Marin-one Medical Devices Co., Ltd. is specialized in manufacturing Orthopedic Implant and instruments, a diversified model of Research and development, production, sales, export. The company has perfect quality management system and advanced production capacity. After more than 18 years research and development, we have about 11 main product series and they are Spinal system, Intramedullary Nail system, Trauma plate and screw system, Locking Plate and screw system, CMF Maxillofacial system, External Fixation, Joint system, Medical Power Tool system, general surgical instruments system, Sterilization Box & basket, veterinary orthopedic etc.
In the principle of "quality first, service first, R&D first, innovation first", the company wins an excellent reputation both in domestic and abroad. Customer satisfaction is the purpose of our service. Since the establishment of the company, all employees have consistently adhered to the business philosophy of "sincere service, continuous improvement", adhered to the service tenet of customer centeredness, pursuit of nobility, attention to details, and reputation first. With professional after-sales service and pioneering spirit, we have established a good reputation.



