Advanced Orthopedic Solutions for Complex Bone Reconstructions
The global orthopedic market is witnessing a paradigm shift toward Hybrid External Fixators. These sophisticated devices, which merge the circular stability of Ilizarov rings with the streamlined ease of unilateral fixators, are becoming the gold standard for treating periarticular fractures, limb lengthening, and non-unions. As a premier manufacturer in China, we recognize that the international demand is driven by an aging population and a rise in high-energy trauma cases resulting from urbanization and transport infrastructure growth across emerging economies.
In North America and Europe, the focus has shifted toward lightweight materials like carbon fiber and high-strength titanium alloys. Meanwhile, in the Asia-Pacific and African markets, there is a burgeoning need for cost-effective yet high-precision surgical instruments. Our facility bridges this gap by providing CE and ISO-certified hybrid systems that offer "Western Quality" with "Global Accessibility."
Utilizing computational biomechanics to simulate load distribution and optimize pin placement for faster bone healing.
Designed for multi-planar stability, allowing early weight-bearing and improving postoperative patient mobility.
Serving over 50 countries with streamlined logistics and localized regulatory compliance support.
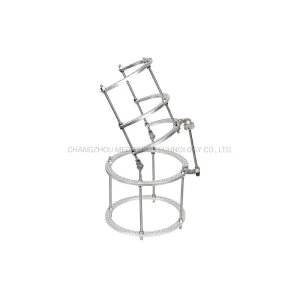
The Ilizarov Ring External Fixator represents the pinnacle of bone segment transport and deformity correction. Our hybrid variations allow surgeons to combine these rings with unilateral rods, providing unmatched flexibility in the operating room. This system is particularly vital for wrist and ankle joint stability where traditional plating might fail due to soft tissue constraints.
Link: Explore Ilizarov Wrist Joint Series
The industry is moving toward "Smart Fixators." We are currently researching the integration of micro-sensors into our Hybrid External Fixators to monitor bone strain and healing progress in real-time. This data can be transmitted to a surgeon's smartphone, allowing for data-driven adjustments to the fixation tension.
Another significant trend is the rise of Minimally Invasive Surgery (MIS). Hybrid fixators facilitate MIS by allowing percutaneous pin insertion, which preserves the blood supply to the bone (the periosteum), leading to significantly faster callus formation. Furthermore, the 3D printing of customized connectors is becoming a reality, allowing for fixators that perfectly match the patient's unique anatomy.
Sustainability is also entering the medical device manufacturing space. Our factory is adopting green manufacturing processes, reducing material waste in the CNC milling of our aluminum components while maintaining the rigorous sterilization standards required for surgical implants.
Years Experience
Export Countries
Product Series
Quality Inspected

Our Ankle Joint External Fixators are specifically engineered for pilon fractures and severe dislocations. By utilizing a hybrid ring-and-bar configuration, we ensure that the joint space is maintained while providing the rigid fixation necessary for bone fusion or healing.
Link: View Ankle Joint Fixation Systems
Global procurement teams favor this model due to its high reuse rate of non-disposable components and the simplicity of its mechanical locking mechanism, which reduces time spent in the surgical theater.
Located in Guangdong, the heart of China's medical tech hub, we have direct access to high-purity raw materials and advanced coating technologies.
Every screw and pin undergoes rigorous stress testing. Our ISO 13485 certification ensures every batch meets international medical standards.
We provide customized external fixation solutions, tailoring lengths and diameters to meet specific regional clinical needs (e.g., pediatric vs. bariatric applications).
International hospital groups and humanitarian organizations (such as the Red Cross) prioritize Hybrid External Fixators that are durable, easy to apply in field conditions, and packaged in sterilized, ready-to-use kits. As an exporter, we optimize our packaging to withstand long-distance shipping and varying humidity levels, ensuring that the instruments arrive in pristine condition.
Guangdong Marin-one Medical Devices Co., Ltd. is specialized in manufacturing Orthopedic Implant and instruments, a diversified model of Research and development, production, sales, export. The company has perfect quality management system and advanced production capacity. After more than 18 years research and development, we have about 11 main product series and they are Spinal system, Intramedullary Nail system, Trauma plate and screw system, Locking Plate and screw system, CMF Maxillofacial system, External Fixation, Joint system, Medical Power Tool system, general surgical instruments system, Sterilization Box & basket, veterinary orthopedic etc.




In the principle of "quality first, service first, R&D first, innovation first", the company wins an excellent reputation both in domestic and abroad. Customer satisfaction is the purpose of our service. Since the establishment of the company, all employees have consistently adhered to the business philosophy of "sincere service, continuous improvement", adhered to the service tenet of customer centeredness, pursuit of nobility, attention to details, and reputation first.