 High Quality CE ISO Certified Cruciate Ligament Circle Titanium Button-Factory Manufactured Endobutton
High Quality CE ISO Certified Cruciate Ligament Circle Titanium Button-Factory Manufactured Endobutton
 Czmeditech Ankle Joint External Fixator Ilizarov Ring External Fixator Orthopedic Instruments
Czmeditech Ankle Joint External Fixator Ilizarov Ring External Fixator Orthopedic Instruments
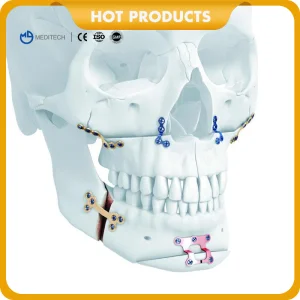 Titanium Cmf Locking Plates and Screws-Maxillofacial Orthopedic Implant System
Titanium Cmf Locking Plates and Screws-Maxillofacial Orthopedic Implant System
 Orthopedic External Fixator - Ilizarov External Fixator-Half Ring-Alluminum Alloy
Orthopedic External Fixator - Ilizarov External Fixator-Half Ring-Alluminum Alloy
 Rail Cast-in Shoulder for Railway Fastening
Rail Cast-in Shoulder for Railway Fastening
 Safe and Reliable Titanium Coating Surgical Instrument Set Orthopedic Implant Plif Lumbar Interbody Fusion Peek Cage System
Safe and Reliable Titanium Coating Surgical Instrument Set Orthopedic Implant Plif Lumbar Interbody Fusion Peek Cage System
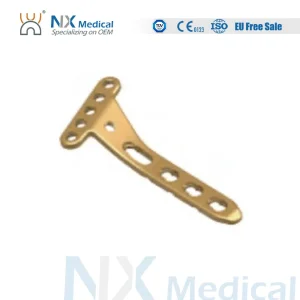 Nx Medical Variable Angle LCP Plate Radius Fragment Locking Mechanism System
Nx Medical Variable Angle LCP Plate Radius Fragment Locking Mechanism System
 Acl Interference Screw for Knee Arthroscopy Peek Material Best Quality Minimally Invasive Orthopedic Implants
Acl Interference Screw for Knee Arthroscopy Peek Material Best Quality Minimally Invasive Orthopedic Implants
The global market for Endobuttons and ACL/PCL fixation has witnessed exponential growth over the last decade, driven by the increasing prevalence of sports-related injuries and a rising aging population that remains active. Cruciate ligament reconstruction is no longer a niche orthopedic procedure but a standard surgical intervention in sports medicine clinics worldwide. From professional athletes in the UEFA leagues to amateur enthusiasts in local gyms, the demand for reliable, biocompatible, and high-strength fixation devices is at an all-time high.
Currently, the market is characterized by a transition from traditional metallic implants to advanced polymer materials like PEEK (Polyetheretherketone) and bio-absorbable composites. North America and Europe remain the largest consumers, but the Asia-Pacific region is emerging as the fastest-growing hub, with China leading the way as both a significant consumer and a powerhouse of orthopedic implant manufacturing. The industrial landscape is shifting towards specialized factories that can offer high-precision CNC machining and clean-room assembly, ensuring that every titanium button or interference screw meets the rigorous standards of global health authorities.
Increasing adoption of minimally invasive arthroscopic surgeries requiring smaller, more robust fixation devices like adjustable loop endobuttons.
Shift towards bio-composite materials that promote osteointegration, reducing the need for secondary surgeries to remove hardware.
Integration of AI-driven quality control and 3D printing for customized surgical guides, enhancing the precision of ACL/PCL placement.

Our flagship Titanium Endobutton is engineered for maximum pull-out strength and surgical simplicity. Designed for femoral fixation in ACL and PCL reconstruction, this device utilizes medical-grade Titanium Alloy (Ti6Al4V ELI) to provide an inert, biocompatible solution that integrates seamlessly with human bone.
As a leading factory, we ensure that every button undergoes rigorous fatigue testing to simulate the stresses of post-operative rehabilitation. Learn more about our Titanium Button solutions.
Procurement departments in modern hospitals and large-scale medical distributors are no longer just looking for the lowest price. They are seeking strategic manufacturing partners who can provide consistency, traceability, and regulatory compliance. The demand for CE and ISO certified ACL/PCL fixation devices has become a baseline requirement for entering the European and Southeast Asian markets.
Localized application scenarios also play a crucial role. For instance, in regions with a high volume of pediatric sports injuries, surgeons require smaller fixation sizes and specialized growth-plate sparing designs. In contrast, in geriatric populations where bone density might be lower, interference screws with deeper thread patterns are preferred to ensure stable graft fixation. As a factory, understanding these nuances allows us to offer tailored OEM/ODM services that cater to specific regional surgical preferences.

For surgeons preferring non-metallic options, our PEEK Interference Screws offer the ideal balance of strength and visibility under MRI/CT. PEEK's modulus of elasticity is close to that of human cortical bone, reducing stress shielding and promoting a more natural healing environment.
These screws are designed for minimally invasive arthroscopic procedures, featuring a self-tapping thread that simplifies the insertion process. Explore our PEEK Fixation Range.
Guangdong Marin-one Medical Devices Co., Ltd. represents the peak of Chinese orthopedic engineering. Why do global distributors choose us over Western counterparts? The answer lies in the combination of industrial scale, R&D agility, and cost-efficiency.
Our facility in Guangdong utilizes the latest Swiss-type lathes and 5-axis machining centers to produce Endobuttons and ACL/PCL fixation systems that are identical in quality to Tier-1 global brands but at a fraction of the cost. This allows healthcare providers to maintain high standards of patient care while managing tight budgets. Furthermore, our location in the Pearl River Delta ensures rapid logistics and a robust supply chain for raw materials like medical-grade Titanium and PEEK.
Guangdong Marin-one Medical Devices Co., Ltd. is specialized in manufacturing Orthopedic Implant and instruments, a diversified model of Research and development, production, sales, export. The company has perfect quality management system and advanced production capacity. After more than 18 years research and development, we have about 11main product series and they are Spinal system, Intramedullary Nail system, Trauma plate and screw system, Locking Plate and screw system, CMF Maxillofacial system, External Fixation, Joint system, Medical Power Tool system, general surgical instruments system, Sterilization Box&basket, veterinary orthopedic etc. In the principle of "quality first, service first, R&D first, innovation first", the company wins an excellent reputation both in domestic and abroad. Customer satisfaction is the purpose of our service.
Since the establishment of the company, all employees have consistently adhered to the business philosophy of "sincere service, continuous improvement", adhered to the service tenet of customer centeredness, pursuit of nobility, attention to details, and reputation first. With professional after-sales service and pioneering spirit, we have established a good reputation.




The success of an ACL or PCL reconstruction surgery is largely dependent on the primary fixation of the graft. Whether using an autograft (from the patient's own body) or an allograft, the Endobutton acts as the anchor point on the femoral side, while interference screws or secondary buttons secure the tibial side. Our manufacturing process focuses on the biomechanical interface—ensuring that the loop material (whether continuous polyester or adjustable) has zero creep and that the button itself has rounded edges to prevent suture fraying.
In the industrial sphere, we are seeing a trend towards "Procedure Packs," where a factory provides not just the implant, but the entire disposable instrument set required for the reconstruction. This reduces the burden of sterilization on hospitals and ensures that every screw is inserted with a perfectly matched driver, minimizing the risk of stripping or surgical complications.
Our commitment to R&D means we are constantly testing new surface treatments, such as HA (Hydroxyapatite) coating on titanium buttons, to further enhance the speed of biological fixation. By staying at the forefront of this technology, Guangdong Marin-one ensures that our partners are always equipped with the latest advancements in orthopedic science.
 Orthopedic External Fixator - Ilizarov External Fixator-C Ring-Alluminum Alloy
Orthopedic External Fixator - Ilizarov External Fixator-C Ring-Alluminum Alloy
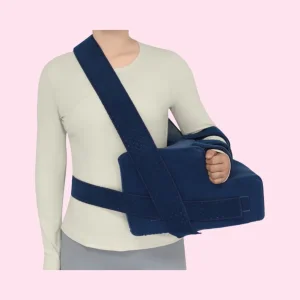 Premium Shoulder Support Pillow for Side Sleepers and Neck Relief of Kmjk
Premium Shoulder Support Pillow for Side Sleepers and Neck Relief of Kmjk
 Carbon Steel/4.8/8.8/10.9 M2.5/Galvanized/Customized Double Rod Fastener Knurled Bamboo Screw
Carbon Steel/4.8/8.8/10.9 M2.5/Galvanized/Customized Double Rod Fastener Knurled Bamboo Screw
 Nx Medical Titanium Medical Orthopedic Trauma Maxillofacial Bone Fracture Reconstruction LCP Locking Compression Plate
Nx Medical Titanium Medical Orthopedic Trauma Maxillofacial Bone Fracture Reconstruction LCP Locking Compression Plate
 Best Price Titanium Coating Surgical Instrument Set Orthopedic Implant Plif Lumbar Interbody Fusion Peek Cage System
Best Price Titanium Coating Surgical Instrument Set Orthopedic Implant Plif Lumbar Interbody Fusion Peek Cage System
 Grab Slotted Headless Plastic Flat Point End Bolt Plla Absorbable Interference Screw Screw
Grab Slotted Headless Plastic Flat Point End Bolt Plla Absorbable Interference Screw Screw
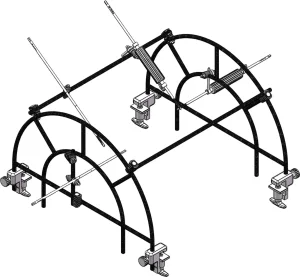 Medical Surgery Orthopedic Pelvic External Fixator Pelvic
Medical Surgery Orthopedic Pelvic External Fixator Pelvic
 Geb242 Replacement Battery for Total Station Ts30 and TM30 Battery
Geb242 Replacement Battery for Total Station Ts30 and TM30 Battery