The global orthopedic market is witnessing a paradigm shift, and at the heart of sports medicine lies the critical demand for high-performance Interference Screws. As a premier Interference Screws Manufacturer & Factory, we recognize that these components are not just fasteners; they are life-changing implants used in Anterior Cruciate Ligament (ACL) and Posterior Cruciate Ligament (PCL) reconstructions. The global commercial status of interference screws is currently valued at billions of dollars, driven by an aging yet active population and a surge in sports-related injuries across North America, Europe, and the Asia-Pacific region.
In today's industrial context, the manufacturing of interference screws has moved beyond simple stainless steel. The industry now demands biocompatible materials such as PEEK (Polyetheretherketone) and Bioabsorbable PLLA/PGA. These materials reduce the risk of secondary surgeries and MRI interference, making them the gold standard in modern clinics. As a leading factory, we leverage Swiss-type CNC machining and injection molding to meet these exacting global standards.
The sports medicine market is projected to grow at a CAGR of 6.5%, significantly boosting demand for fixation devices.
Integration of bioactive glass and hydroxyapatite into bioabsorbable screws for faster bone integration.
Sub-micron tolerances ensure the screw thread provides maximum pull-out strength in cancellous bone.

Our flagship Bioabsorbable Interference Screw is designed for surgeons who prioritize long-term patient recovery. By utilizing high-grade PLLA/PGA blends, this screw provides rigid fixation during the critical healing phase (6-12 months) and gradually resorbs, leaving only natural bone behind.
Modern procurement in the medical field is no longer just about price; it is about regulatory compliance and material traceability. Hospitals and surgical centers worldwide are increasingly seeking Interference Screws Manufacturers that offer CE, ISO 13485, and FDA-compliant production lines. There is a visible trend toward "Minimally Invasive Surgery" (MIS), where screws must be cannulated to allow for guide-wire placement, ensuring absolute precision in tight joint spaces.
Furthermore, localized application scenarios vary. In high-income regions, there is a heavy preference for PEEK Interference Screws due to their radiolucency, allowing for clear post-operative imaging. In emerging markets, there is a massive demand for high-quality, cost-effective titanium and stainless steel alternatives for general trauma and orthopedic clinics. As a global factory, we cater to both ends of the spectrum, providing customized solutions for diverse surgical requirements.
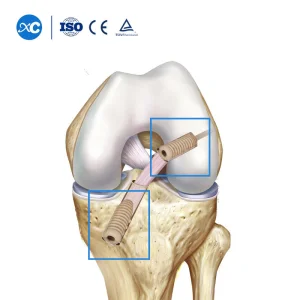
Our Non-Absorbable Cannulated PEEK Interference Screws represent the pinnacle of polymer technology. PEEK offers a modulus of elasticity similar to human bone, reducing stress shielding and promoting a healthier biological environment for ligament-to-bone healing.
Choosing a Chinese factory for your orthopedic implant supply chain offers unparalleled strategic advantages. Our facility in Guangdong integrates the entire lifecycle of medical device production. We offer OEM and ODM services that allow global brands to customize screw dimensions, thread pitches, and drive types (e.g., Hex, Star, or Torx) according to their specific surgical protocols.
Our advantages include:
Scaling production allows us to offer competitive pricing without compromising on the medical-grade quality of raw materials like PEEK and Titanium.
Every single interference screw undergoes rigorous testing, including torque testing, pull-out strength analysis, and sterility verification.
Our AI-driven manufacturing processes enable us to transition from a surgical concept to a physical medical device faster than traditional European factories.
Guangdong Marin-one Medical Devices Co., Ltd. is specialized in manufacturing Orthopedic Implant and instruments, a diversified model of Research and development, production, sales, export. The company has perfect quality management system and advanced production capacity. After more than 18 years research and development, we have about 11 main product series and they are Spinal system, Intramedullary Nail system, Trauma plate and screw system, Locking Plate and screw system, CMF Maxillofacial system, External Fixation, Joint system, Medical Power Tool system, general surgical instruments system, Sterilization Box & basket, veterinary orthopedic etc. In the principle of "quality first, service first, R&D first, innovation first", the company wins an excellent reputation both in domestic and abroad. Customer satisfaction is the purpose of our service.
Since the establishment of the company, all employees have consistently adhered to the business philosophy of "sincere service, continuous improvement", adhered to the service tenet of customer centeredness, pursuit of nobility, attention to details, and reputation first. With professional after-sales service and pioneering spirit, we have established a good reputation.



