












| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
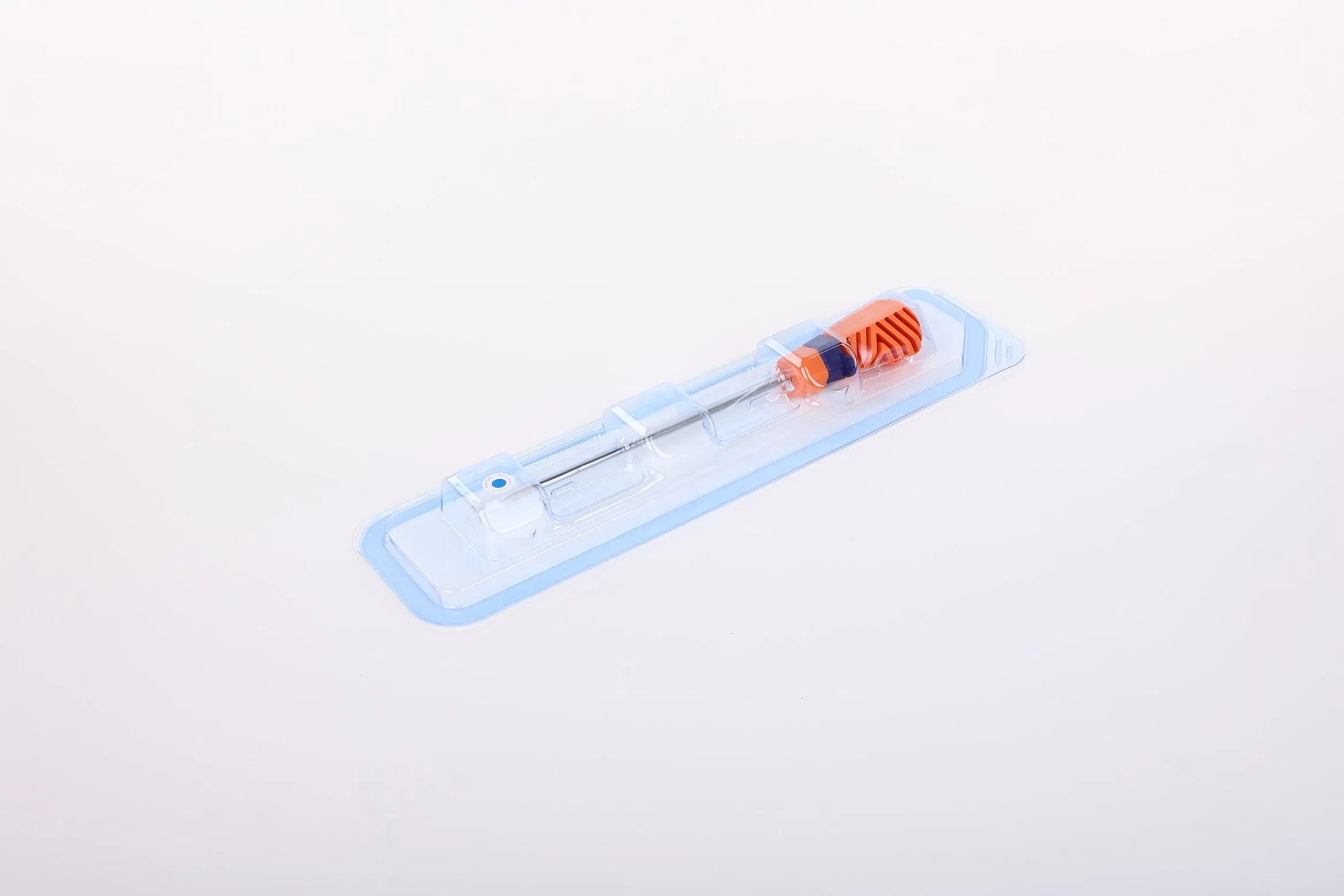
| Model NO. | WZ-MDT-D-05 |
| Certification | CE, ISO13485 |
| Usage | Orthopedic Surgery |
| Color | Silver and Orange |
| Function | Connect The Soft Tissue and Bone |
| Specifications | Peek, Titanium Screw |
| Sizes Available | 1.8mm, 2.8mm, 3.5mm, 4.0mm, 4.5mm, 5.0mm, 5.5mm, 6.5mm |
| Sterility | Eo Sterile |
| Warranty | 3 Years |



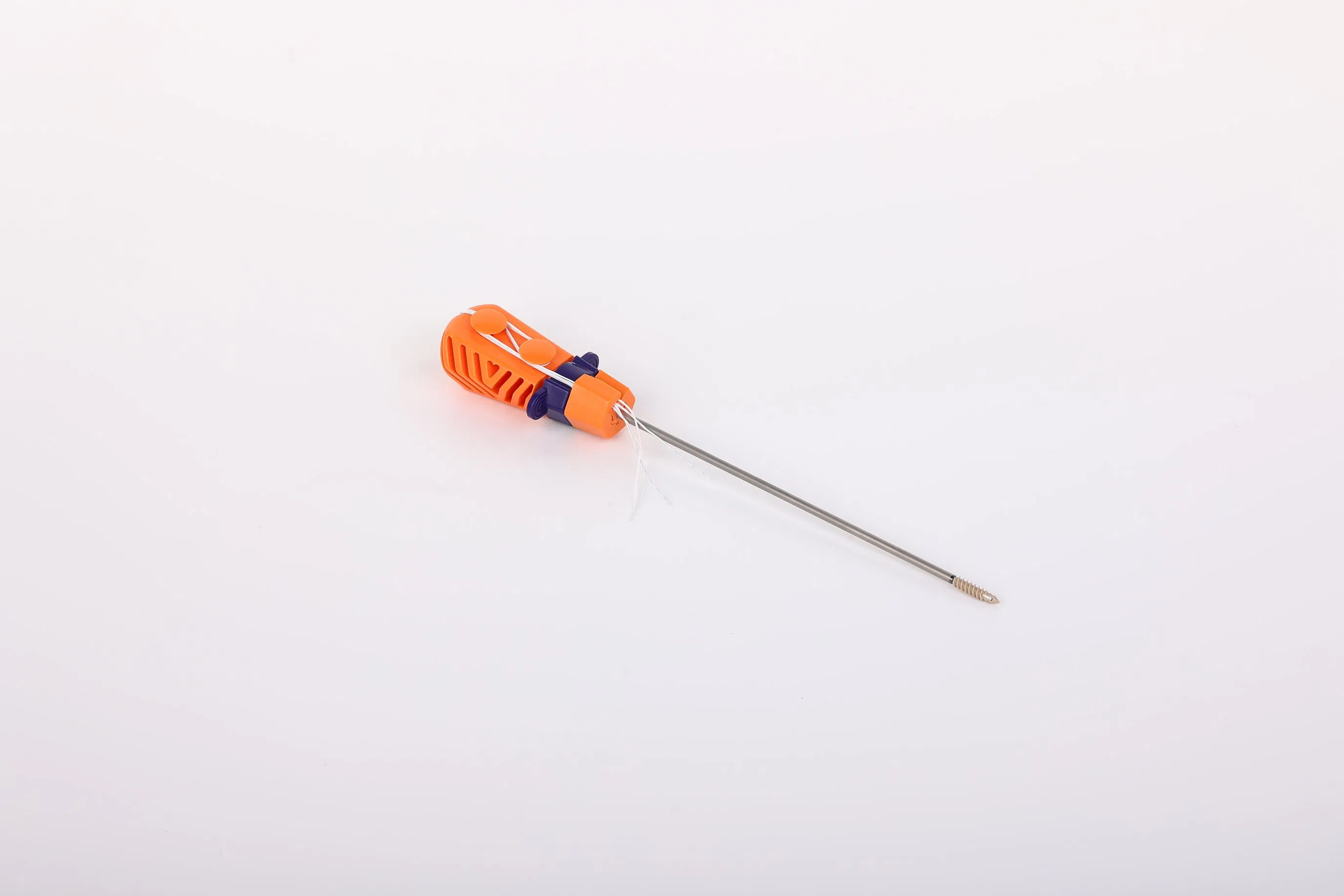
Anchor Material: Made of Ti6Al4V alloy, conforming to ISO5832-3:2016 requirements, ensuring high biocompatibility and strength for orthopedic applications.
Suture Material: Crafted from ultra-high molecular weight polyethylene (UHMWPE) without coating. It is woven from several UHMWPE yarns, meeting ASTM F2848-17 standards. The blue suture is dyed with FDA-approved materials for clear visibility during surgery.
Inserter Design: The portion contacting the human body is constructed from stainless steel (ASTM F899-12b). The handle is ergonomically designed using polycarbonate and ABS materials for a non-slip, precise grip.
Our comprehensive product range includes Disposable Surgical Lavage Systems, Suture Anchor Systems, Meniscal Repair Systems, Shaver Blades, Bone Cement Mixers, Rib Splints, and Finger Splints. With advanced production capabilities, these orthopedic surgical consumables are used in more than 40 countries globally.


To guarantee product quality and safety, investments have been made in Class 100,000 clean workshops and Class 10,000 purification laboratories, supporting full-cycle capabilities in R&D, manufacturing, and sales.

 Marin-one Medical
Marin-one Medical