







| Customization: | Available |
|---|---|
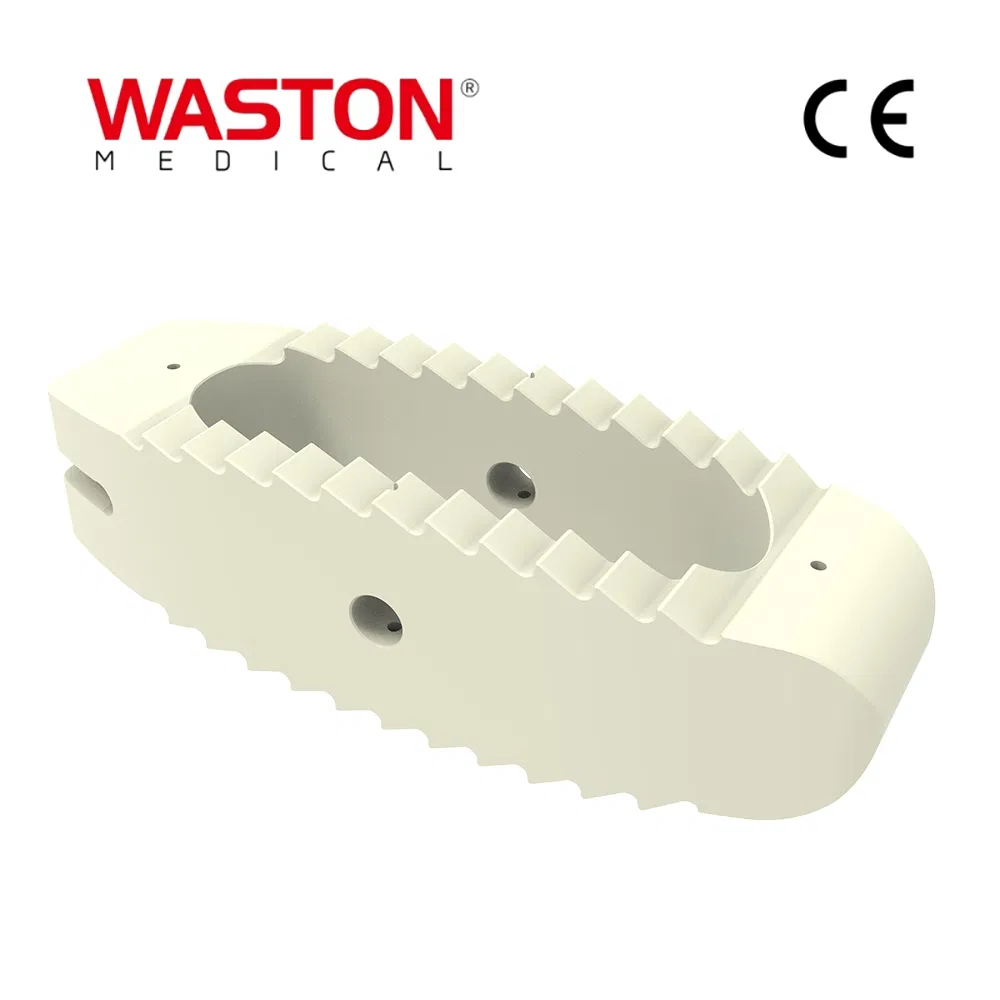
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
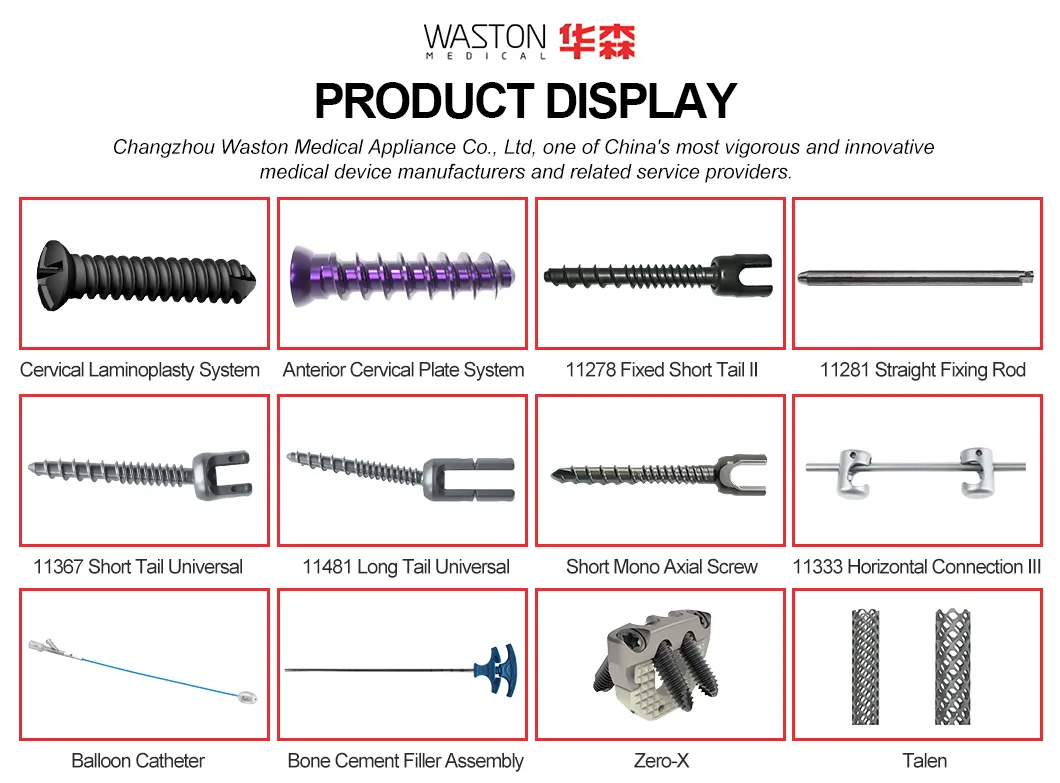
| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | L=8 | ||
| 10511310 | L=10 |


As a national high-tech enterprise, the R&D investment exceeds 8% of total sales annually. We specialize in transforming clinical expertise into innovative medical solutions with national invention patents. Our revolutionary technologies include the Rev Drill system, Three-Row-Stapler, Sternal Fixation, and Rib plate.
Equipped with German and American-imported machining centers and Japanese-imported automatic lathes, we ensure first-class production quality. Our testing center includes a biomechanical laboratory and a physical-chemical testing center for rigorous raw material control and performance analysis.

Our orthopaedic implants are exported to numerous countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. Based on global feedback, our products consistently meet and exceed international medical standards.



 Marin-one Medical
Marin-one Medical