

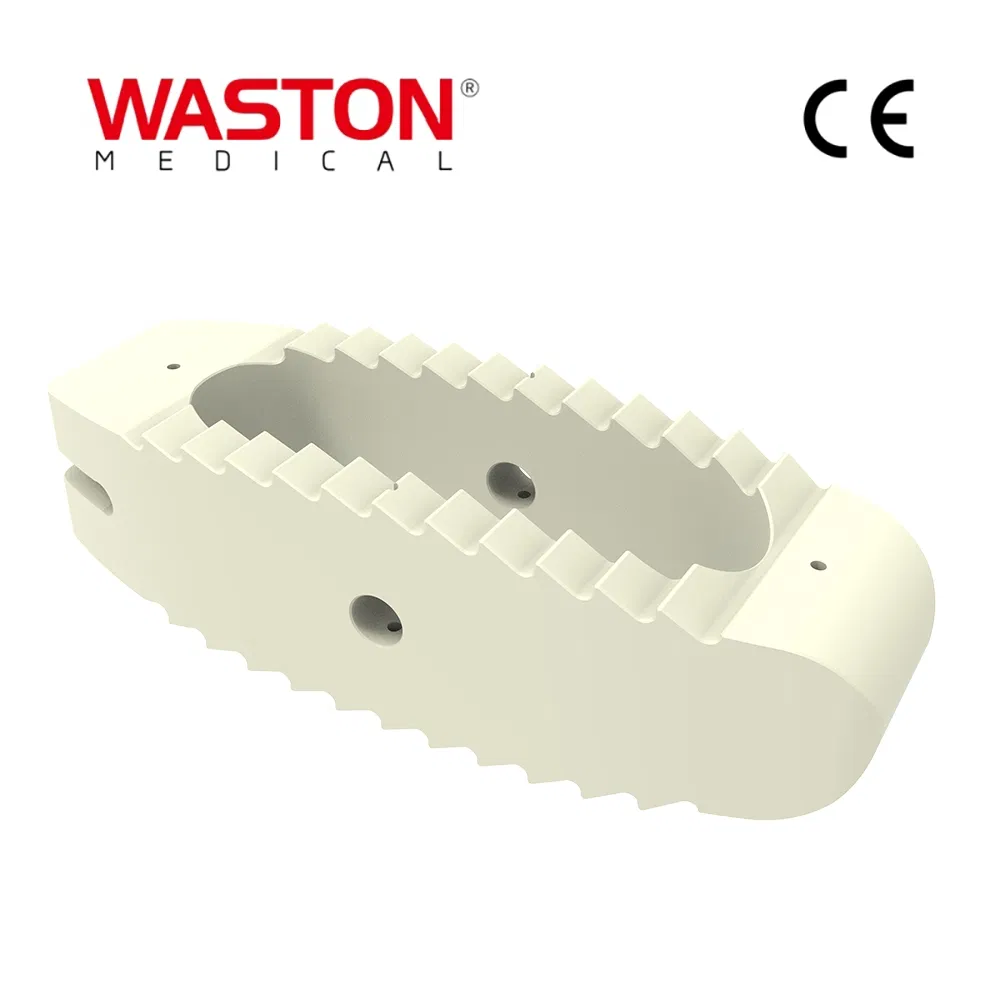






| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
| Model NO. | Cage System | Material | Pure Titanium/Titanium Alloy/Stainless Steel |
| Feature | Disposable | Certification | CE, ISO, SGS |
| Group | Adult | HS Code | 9021100000 |
| Transport Package | Non-Sterile Package, Paper Plastic Roll Bag | ||

| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| NEULEN Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| NEULEN Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | φ2.5 | L=8 | |
| 10511310 | φ2.5 | L=10 |

As a national high-tech enterprise, the proportion of R&D investment to total sales has been over 8% for consecutive years. We focus on transforming clinical experience into innovative achievements, holding multiple national invention patents.


Our facility operates strictly according to the ISO13485 quality system and CE MDD 93/42/EEC directives. We utilize German and American-imported machining centers and Japanese-imported automatic lathes to ensure first-class quality. Our Testing Center includes a biomechanical laboratory and a physical-chemical testing center for rigorous quality control.
Our products are exported to many countries, including France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier to some of the largest global players in the orthopaedic industry.



Yes, we support customized designs based on specific surgical requirements or drawings.
For any verified defective goods, please provide photographic evidence, and we will arrange for replacements.
Yes, we welcome sample orders so you can evaluate our product quality and service in your local market.
Samples are generally delivered within 7 days, while standard orders are processed within 25 days.
Our products are certified with CE and ISO. Additionally, our Spinal Systems carry FDA registration.
We generally maintain stock for listed products. However, availability depends on current sales; please contact our team for real-time inventory updates.
 Marin-one Medical
Marin-one Medical