Basic Information
Model NO.Cage System
MaterialPure Titanium/Titanium Alloy/Stainless Steel
FeatureDisposable
CertificationCE, ISO
GroupAdult
HS Code9021100000
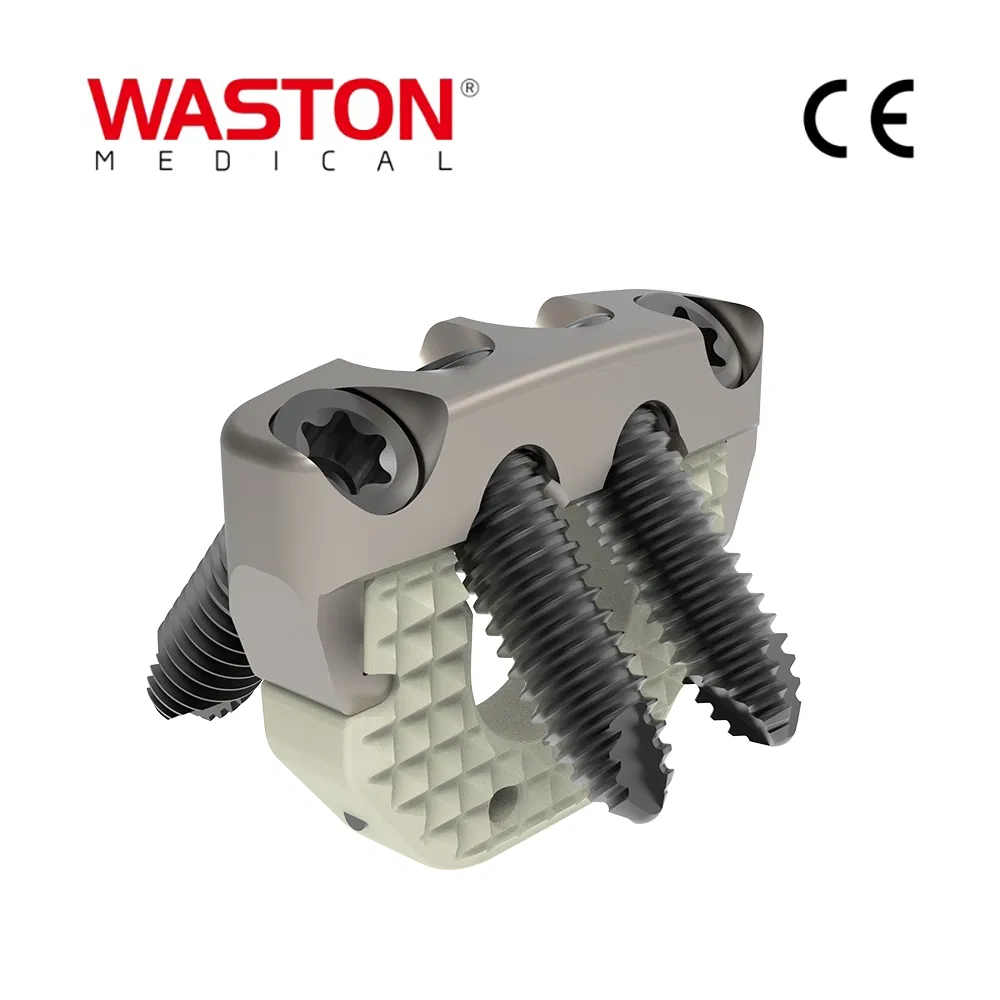
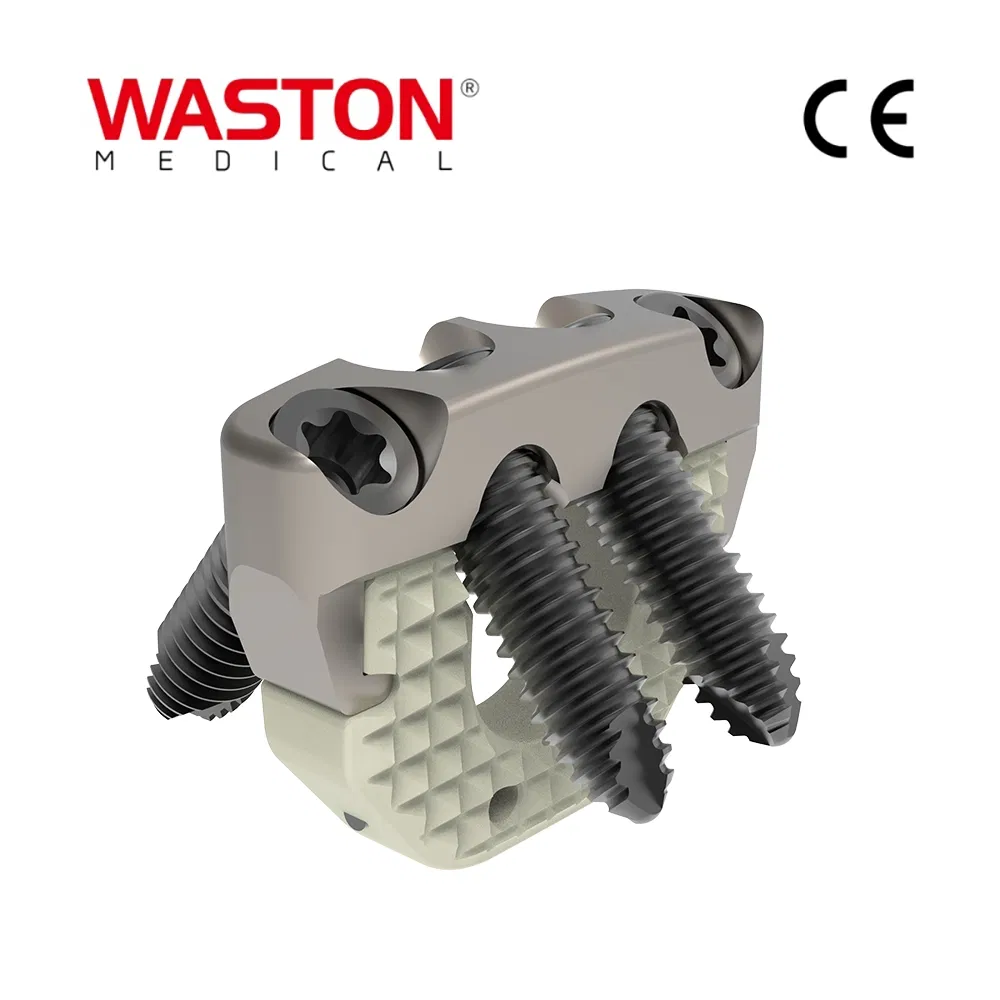
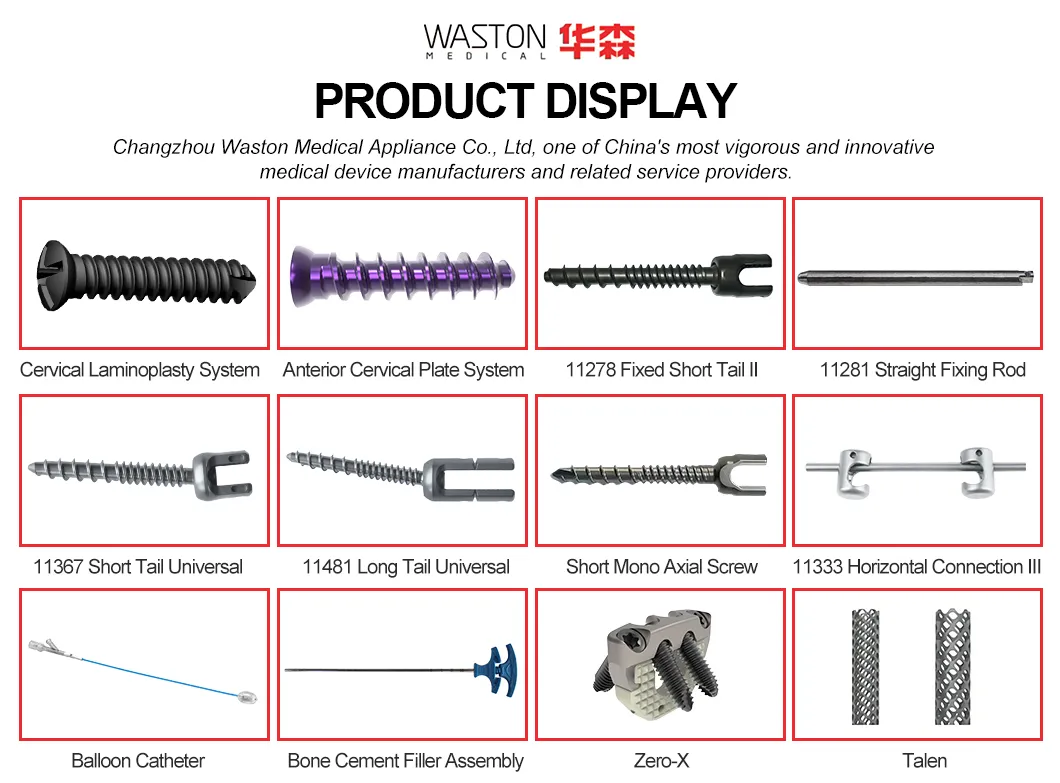
Product Introduction
Main Features
- Immediate lamina supporting in surgery, best maintain the canal plasty expanded.
- The lower dentate supporting plate provides a more stable supporting force.
- Arc openings provide better inclusion.
- Has certain plasticity, can be slightly moulded in operation.
Key Advantages
- Provide a stable support for vertebral body decompression.
- Four holes configuration optimized for surgery requirements.
- Unique angle design ensures vertebral body stability.
Product Specifications
| PRODUCT NAME |
PRODUCT CODE |
DIA |
DATA |
| Laminoplasty Plate |
11101012 |
/ |
H=12 |
| 11101014 |
/ |
H=14 |
| 11101016 |
/ |
H=16 |
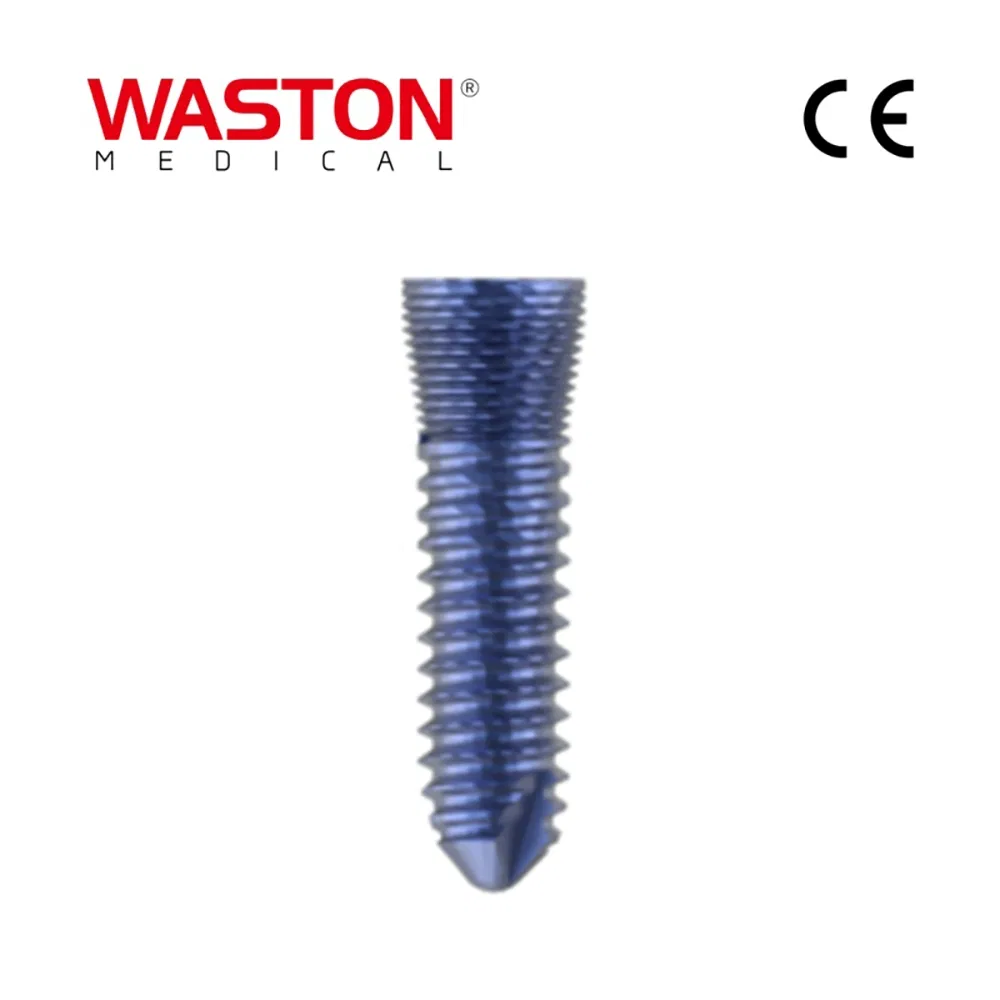
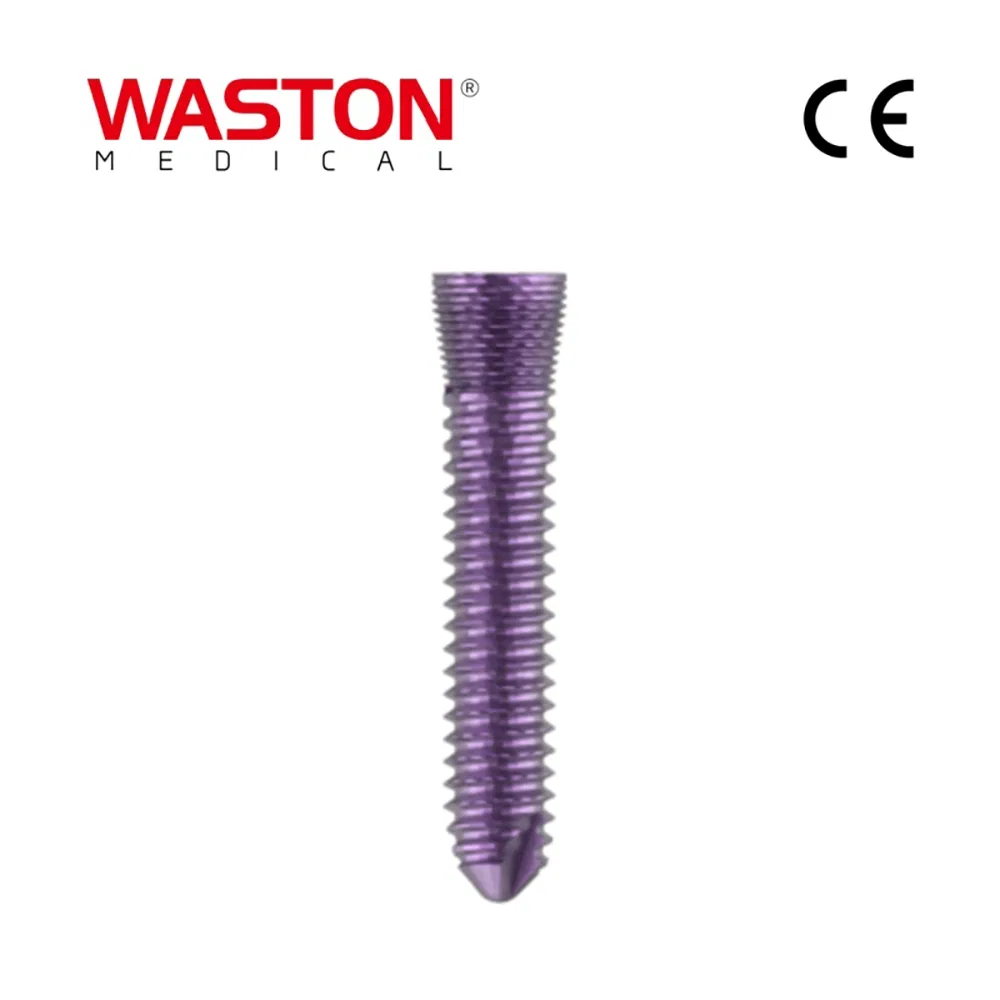
| Screw (Self-tapping) |
10511306 |
φ2.5 |
L=6 |
| 10511308 |
φ2.5 |
L=8 |
| 10511310 |
φ2.5 |
L=10 |
Manufacturing Excellence
As a national high-tech enterprise, R&D investment exceeds 8% of total sales annually. We specialize in transforming clinical experience into innovative medical solutions with national invention patents. Our revolutionary systems like the Rev Drill, Sternal Fixation, and Rib plate are recognized benchmarks in the domestic medical devices industry.
Operating strictly under ISO13485 and CE MDD 93/42/EEC standards, we utilize advanced German, American, and Japanese machining centers to ensure world-class quality. Our testing center includes biomechanical laboratories and physical-chemical testing to control raw materials and product performance.
Certifications & Global Presence
Our products are exported globally to countries including France, Turkey, Russia, and Thailand, meeting diverse international standards. We also serve as a major OEM supplier for global players in the orthopaedic industry.
Packaging & Shipping
Frequently Asked Questions
Q1: Is customized design available for these implants?
Yes, we provide customized design services based on specific requirements or drawings.
Q2: What certificates do your products hold?
We hold CE and ISO certifications, and FDA approval specifically for our Spinal Systems.
Q3: Can I request samples for quality testing?
Yes, we welcome sample orders so you can test our quality and market compatibility in advance.
Q4: What is the typical delivery time for orders?
Samples are usually delivered within 7 days, while bulk orders typically take within 25 days.
Q5: Do you maintain stock for regular items?
Generally, we keep stock for listed standard products, though it is best to confirm current inventory levels with our team.
Q6: What is your policy for defective goods?
If goods are found to be defective, please provide photo documentation for confirmation, and we will arrange for replacements.















 Marin-one Medical
Marin-one Medical