About Us
Established in 1958, we are specialized in the development, manufacture, and sales of orthopedic implants and instruments. As a leading enterprise in the medical apparatus industry, our comprehensive product line covers spine pedicle screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, hip prosthesis, and specialized surgical instruments.
Our facilities feature advanced imported CNC machinery, high-precision digital control systems, and a 100,000 Grade sterile purification chamber to ensure superior product quality. We were among the first in the industry to achieve ISO 9001 quality system certification. Our commitment to innovation and quality has earned us numerous national and provincial awards for science and technology.
Frequently Asked Questions
What are your main orthopedic product lines?
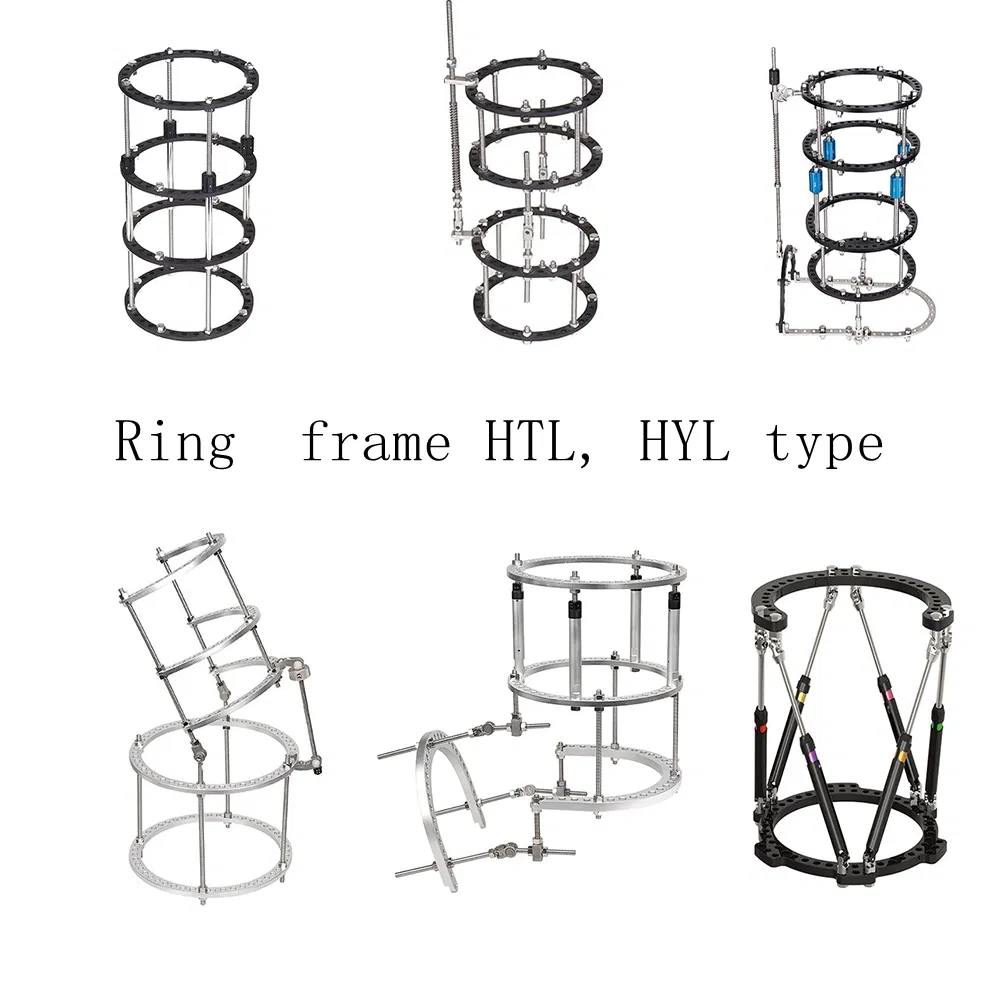
We specialize in trauma plates, interlocking nails, spinal fixators, external fixators, and high-performance power tools for surgical use.
How do you ensure the quality of your medical devices?
All products are manufactured according to international CE and ISO standards. We implement a strict quality control process including pre-production samples and a 100% final inspection before shipment.
Do you offer OEM or ODM services?
Yes, we accept both OEM and ODM orders to meet the specific requirements and designs of our global partners.
What materials are typically used for your fixators?
Our external fixators and implants are primarily crafted from high-grade Aluminum and Stainless Steel to ensure durability and biocompatibility.
What are your accepted payment and delivery terms?
We accept various delivery terms including FOB, CIF, EXW, and Express Delivery. Payments can be made via T/T, L/C, Western Union, or Escrow in USD, EUR, or CNY.
How long has your company been in the orthopedic industry?
We have over 60 years of experience in the R&D, manufacture, and distribution of orthopedic implants since our establishment in 1958.





















 Marin-one Medical
Marin-one Medical