




1 / 5










| Still deciding? Get samples of $ ! US$ 258/Set Request Sample |
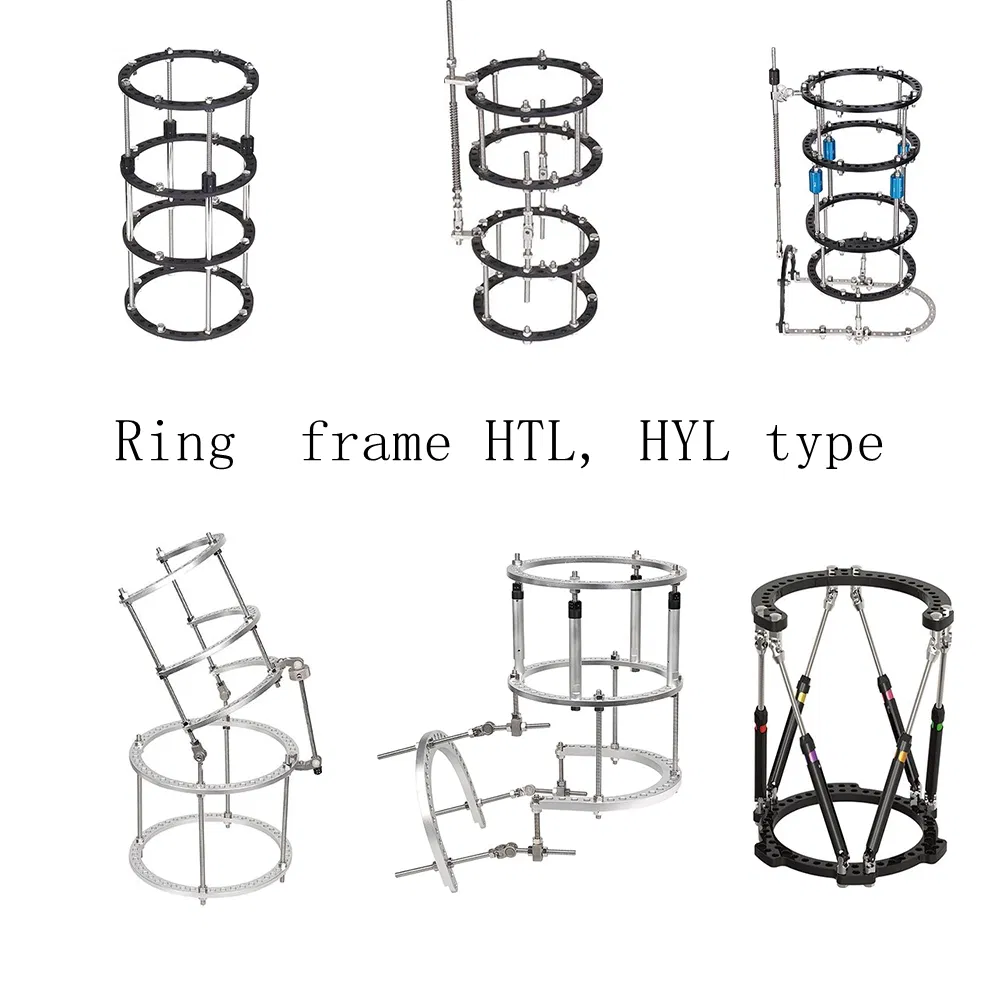
| Product Name | Model | Assembly Part Number | Assembly Part Name | Qty |
|---|---|---|---|---|
| Lower limb lengthening external fixator | HYL003 | HYLL0108-0126 | Rings | 4 |
| HYLY0108-0112 | Inner threaded connecting rod | 4 | ||
| HYLL05 | Thread rod (M6*150) | 4 | ||
| HYLL05 | Thread rod (M6*120) | 4 | ||
| HYLJ01 | Pin clamp | 16 |






 Marin-one Medical
Marin-one Medical