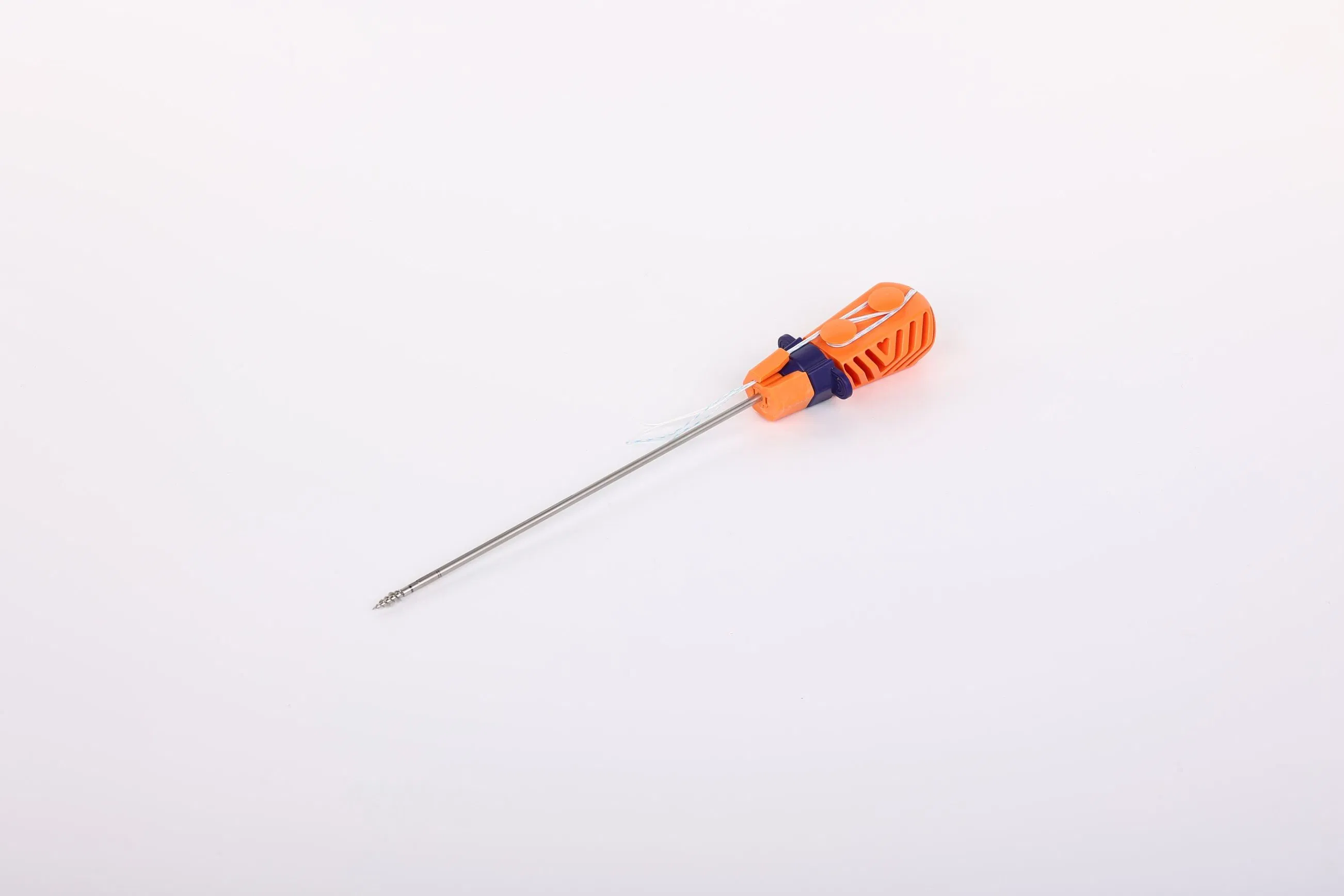
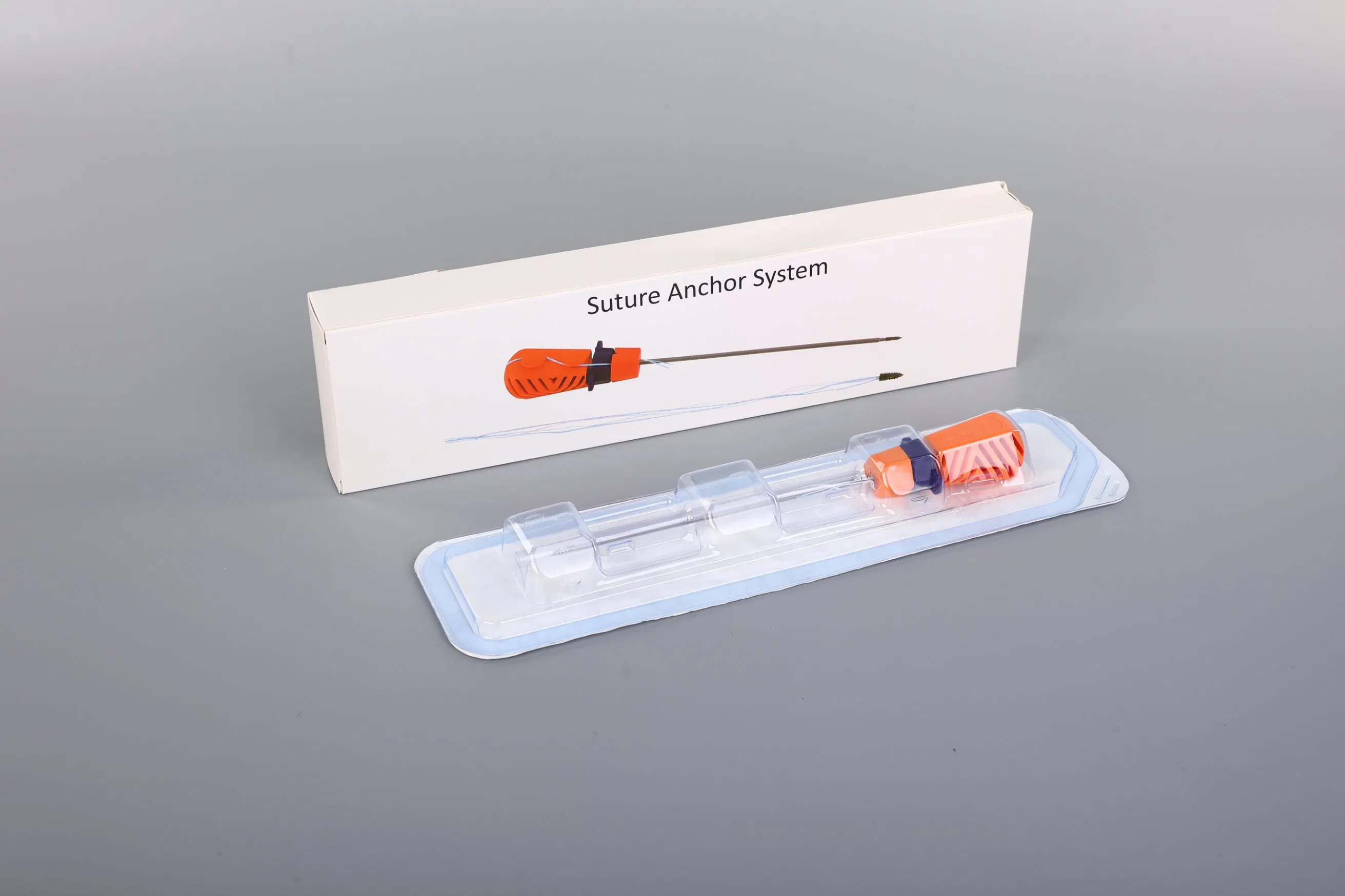
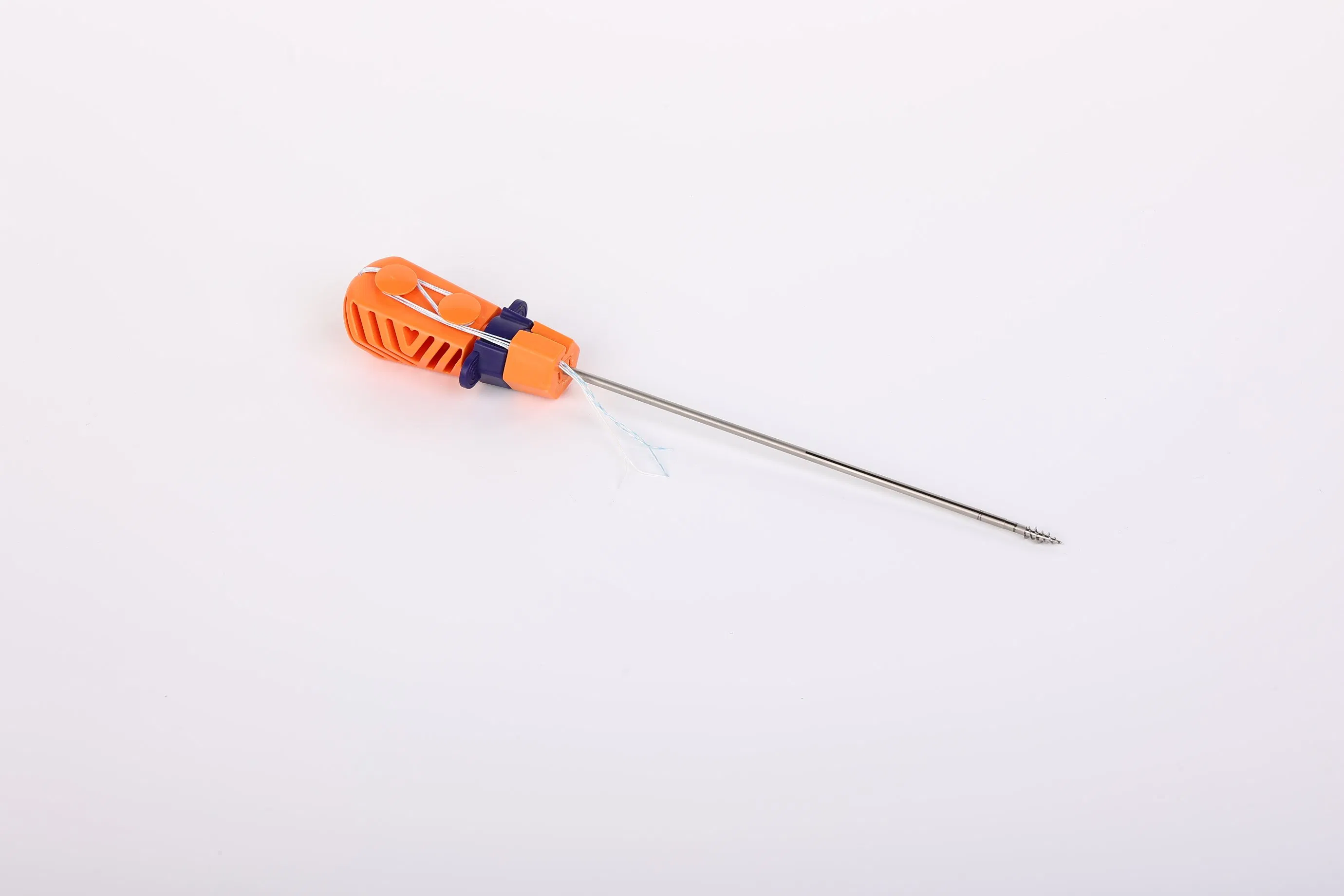
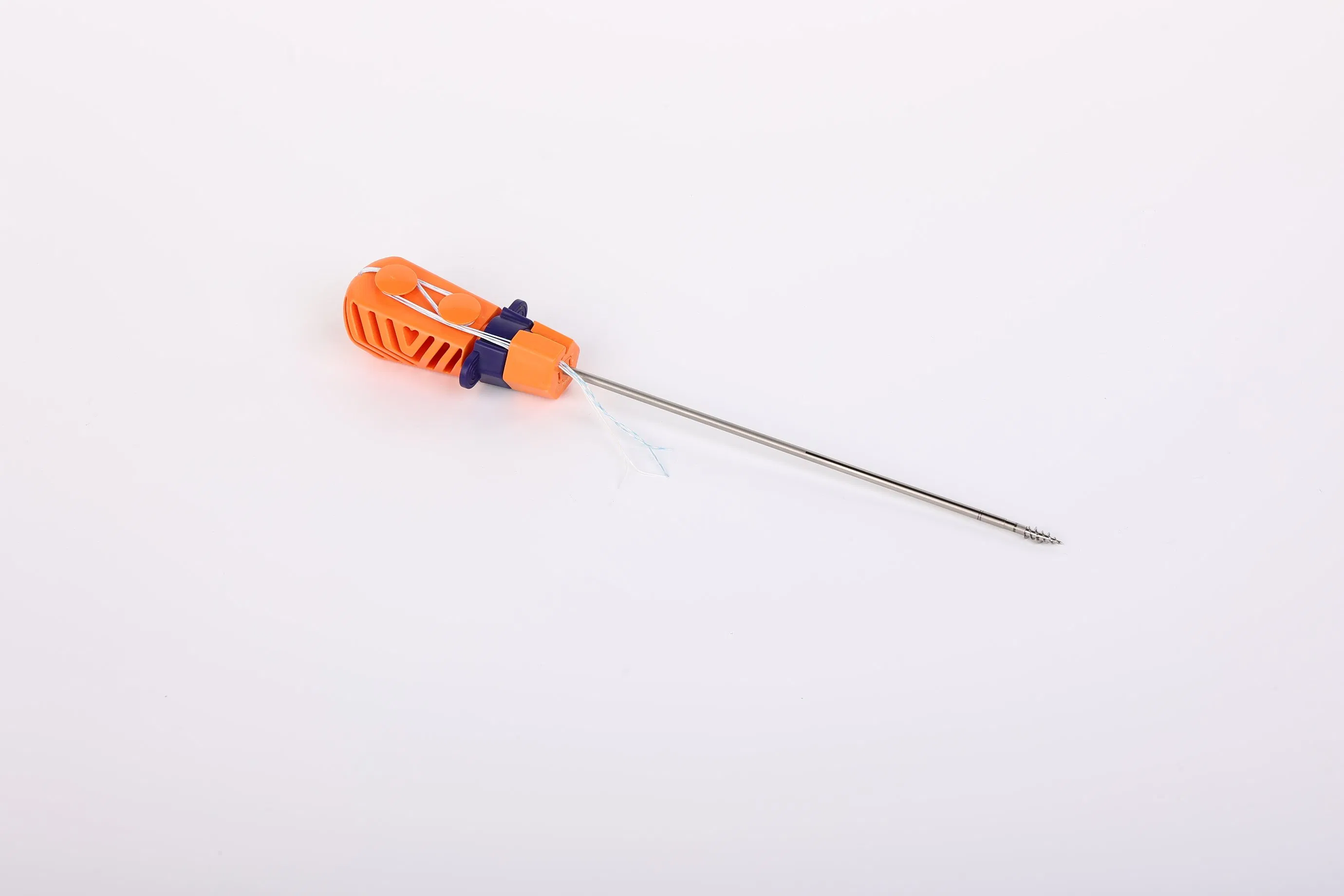
The Suture Anchor System is a high-precision orthopedic solution consisting of an anchor, non-absorbable suture, and an inserter, specifically designed for joint repair surgeries.
- Anchor Material: Crafted from Ti6Al4V alloy, strictly meeting ISO 5832-3:2016 standards for biocompatibility and strength.
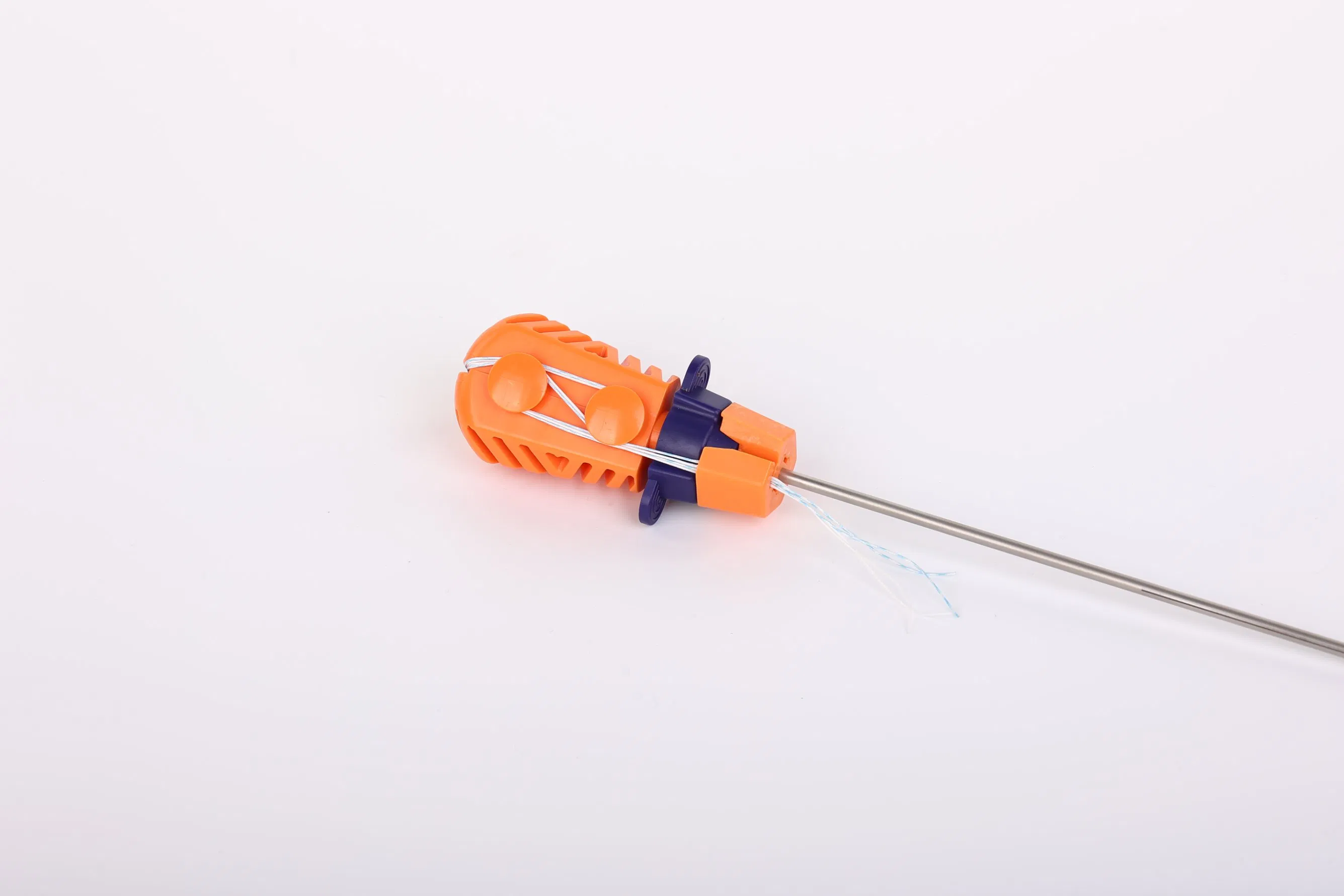
- Advanced Suture: Utilizes ultra-high molecular weight polyethylene (UHMWPE) yarn. The suture is woven for maximum durability and meets ASTM F2848-17 requirements.
- Safe Coloration: Blue sutures are dyed with FDA-approved materials (code 73.1015) for clear visibility during surgery.
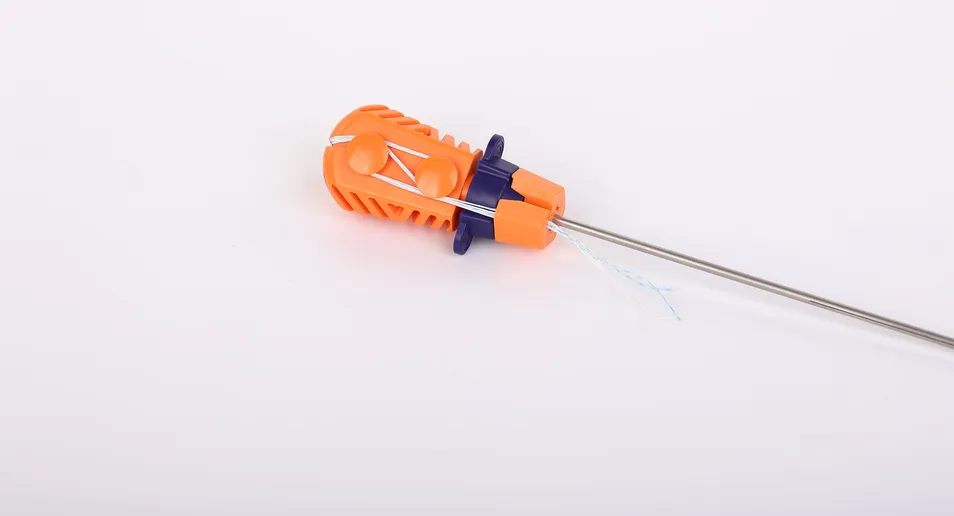
- Ergonomic Inserter: The contact parts are made of surgical-grade stainless steel (ASTM F899), while the handle is designed with high-quality polycarbonate and ABS for easy handling.
Our production process ensures the highest safety standards for orthopedic surgical consumables. We maintain advanced production capabilities to serve global medical institutions across more than 40 countries.
- Controlled Environment: All products are manufactured in Class 100,000 clean workshops.
- Laboratory Testing: Supported by Class 10,000 purification laboratories for R&D and quality verification.
- Global Standards: Fully compliant with CE certification and ISO 13485 quality management systems.
🟠 Frequently Asked Questions
Are you a manufacturer or a trading company?
We are a professional manufacturer with dedicated production facilities, clean workshops, and an in-house R&D team specialized in orthopedic instruments.
What materials are used in the Suture Anchor System?
The anchors are made of medical-grade Ti6Al4V alloy or PEEK, and the sutures are made of Ultra High Molecular Weight Polyethylene (UHMWPE) for superior strength.
Is OEM service available for custom requirements?
Yes, we provide flexible OEM services, including custom designs and tailored packaging solutions to align with your specific brand requirements.
What certifications do your products carry?
All our orthopedic surgical consumables are fully compliant with CE certification and ISO 13485 international quality standards.
How is the product sterilized and what is the warranty?
The products are EO (Ethylene Oxide) sterilized for safety. We provide a 3-year warranty for this system.
Can I request a sample before a bulk order?
Yes, samples can be provided for quality evaluation and testing. Please contact our sales team to arrange details.
















 Marin-one Medical
Marin-one Medical