
Cannulated Titanium Stainless Steel Tibia Nail Intramedullary Nails with 4.5 Locking Screws

High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors

Surgical Instrument Titanium Gamma Femoral Intramedullary Nails

Orthopedic PFNA Femoral Implant PFN Proximal Femur Intramedullary Interlocking Nail
The United Kingdom has long been at the forefront of surgical innovation, particularly in the field of orthopedics. From the pioneering work of Sir John Charnley to the modern-day advancements in the National Health Service (NHS), the demand for high-quality Intramedullary (IM) Nails has grown exponentially. As a dedicated manufacturer and exporter to the United Kingdom, we understand that the UK market is characterized by a rigorous adherence to safety standards, including the post-Brexit UKCA marking and traditional CE certification.
In cities like Sheffield, often referred to as the "Steel City" with a rich history in medical device manufacturing, and the industrial hubs of the Midlands, the standard for surgical steel and titanium is exceptionally high. Our Intramedullary Nails are engineered to meet these British standards, ensuring that surgeons in London, Manchester, and Glasgow have access to implants that provide optimal stability, early weight-bearing, and reduced complications for patients suffering from long bone fractures.
Compliance with the NHS Supply Chain requirements ensures our products offer both clinical efficacy and cost-effectiveness for the public health sector.
Our manufacturing processes are strictly aligned with ISO 13485 standards and prepared for the evolving UK medical device regulations.
Utilizing high-grade Titanium Alloy (Ti-6Al-4V ELI) which is a staple in the UK's high-end orthopedic surgeries for biocompatibility.
The future of orthopedic implants in the United Kingdom is increasingly digital. We are witnessing a shift toward AI-assisted surgical planning and 3D-modeled implants. Our production facility integrates AI-driven CNC machining to ensure that every intramedullary nail—whether for the femur, tibia, or humerus—features micron-level precision. This technological synergy is vital as UK trauma centers move toward Minimally Invasive Surgery (MIS) techniques to reduce hospital stay times and improve patient throughput in a busy NHS environment.
UK Market Trend: There is a significant move toward "Intertan" style nails for geriatric hip fractures, a major focus for the British Orthopaedic Association (BOA). Our PFNA and Gamma nail systems are designed to address the specific needs of the UK's aging population, providing superior "cut-out" resistance and rotational stability.
Beyond the hardware, the "exporter" role involves sophisticated logistics. We provide seamless door-to-door delivery to major UK ports and distribution centers, ensuring that whether a hospital is in rural Cornwall or the heart of London, the supply of critical trauma implants remains uninterrupted.
In the UK, Major Trauma Centres (MTCs) like the Royal London Hospital or the Queen Elizabeth Hospital Birmingham require immediate access to comprehensive IM nailing systems. Our products are utilized in various high-stakes scenarios:
By focusing on the "Total Solution" approach—providing not just the nail, but also the precision reamers, guide wires, and locking screw sets—we support British surgeons in achieving the best possible clinical outcomes.
Guangdong Marin-one Medical Devices Co., Ltd. is specialized in manufacturing Orthopedic Implant and instruments, a diversified model of Research and development, production, sales, export. The company has perfect quality management system and advanced production capacity. After more than 18 years research and development, we have about 11 main product series and they are Spinal system, Intramedullary Nail system, Trauma plate and screw system, Locking Plate and screw system, CMF Maxillofacial system, External Fixation, Joint system, Medical Power Tool system, general surgical instruments system, Sterilization Box & basket, veterinary orthopedic etc. In the principle of "quality first, service first, R&D first, innovation first", the company wins an excellent reputation both in domestic and abroad. Customer satisfaction is the purpose of our service.
Since the establishment of the company, all employees have consistently adhered to the business philosophy of "sincere service, continuous improvement", adhered to the service tenet of customer centeredness, pursuit of nobility, attention to details, and reputation first. With professional after-sales service and pioneering spirit, we have established a good reputation in the global and UK medical markets.





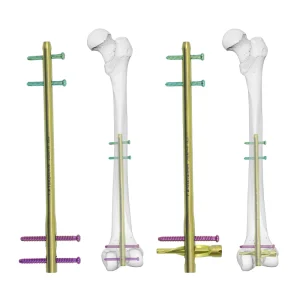
Medical Supply Orthopedic Implant Titanium Dfn Distal Femoral Intramedullary Nail


Factory Instrument Expert Femur Intramedullary Nail Orthopedic Intramedullary Nails for Human
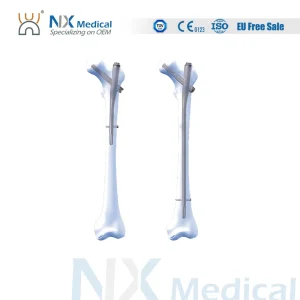
Nx Medical Orthopedic Implant Proximal Femoral Interlocking Intramedullary Nail

Medical Titanium Bone Surgical System Pfna Orthopedic Femoral Interlocking Intramedullary Nail

Orthopedic Implant Interlocking Nail Standard Intertan Intramedullary Nail for Fracture Surgery
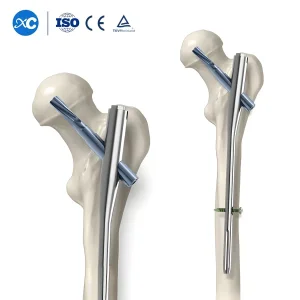
Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail

Intramedullary Nail System Expert Humeral Proximal Interlocking Nail Blade Locking
Whether you are a medical device distributor in London, a surgical consultant in Edinburgh, or a procurement officer for a large NHS Trust, Guangdong Marin-one Medical is ready to support your trauma care needs. Our dedicated UK export team ensures rapid communication, compliance support, and logistics excellence.
Send Inquiry Now