Frequently Asked Questions
What materials are used in the External Fixator system?
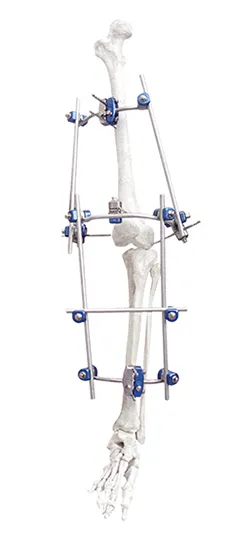
The system utilizes high-grade Titanium Alloy and Stainless Steel for connectors and pins, while the rods are crafted from medical-grade Carbon Fibre for optimal radiolucency and strength.
Is the External Fixator system reusable?
Yes, the core components of the External Fixator system are designed to be reusable after proper sterilization, following standard medical protocols.
What are the benefits of the snap-fit design?
The revolutionary snap-fit design allows for rapid assembly and adjustment during surgery, significantly reducing procedure time while maintaining a secure and stable lock.
Does the system provide good visualization under X-ray?
Yes, by using premium medical-grade aluminum and carbon fiber rods, the system enhances fracture visualization and diagnostic accuracy under imaging equipment.
Can the bone screws be arranged independently?
Absolutely. The modular design allows for the independent arrangement of bone screws for both proximal and distal fractures, providing surgeons with unparalleled customization.
Which certifications does this medical device hold?
Our External Fixation systems are fully certified with CE and ISO13485, ensuring they meet strict international quality and safety standards for medical devices.






















 Marin-one Medical
Marin-one Medical