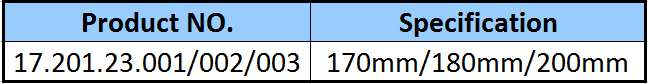
✓
Indication: Ideal for lower limbs fracture treatment and bone lengthening procedures.
✓
Application: Specifically designed for fractures relevant to the articular surface. Utilizes a 4.8mm drill bit and can be used with olive-head Kirschner wires as required.
✓
Screw Compatibility: Supports 6.0/5.0 screws in both Stainless Steel (SS) and Titanium Alloy (TA).
As a national high-tech enterprise, R&D investment remains a priority, transforming clinical expertise into innovative medical achievements. Key proprietary technologies include the Rev Drill system, Three-Row-Stapler, Sternal Fixation, and Rib plates.
Operations strictly adhere to ISO13485 quality systems and CE MDD 93/42/EEC directives. The facility utilizes premium equipment including machining centers imported from Germany and the USA, alongside Japanese automatic lathes.
Products are exported worldwide to regions including Europe, Southeast Asia, and Latin America. The quality consistently meets international standards based on extensive clinical feedback. We also serve as a major OEM supplier for leading global players in the medical device industry.
Frequently Asked Questions
Q1: What are the primary indications for the Multi-ring Combined Fixator?
It is primarily used for treating lower limb fractures and for bone lengthening procedures, especially those involving the articular surface.
Q2: Which materials are available for this fixator system?
The system is available in Pure Titanium, Titanium Alloy, and Stainless Steel to meet different clinical requirements.
Q3: Does the product carry international certifications?
Yes, the products are fully certified with CE and ISO13485 standards, ensuring compliance with international medical safety regulations.
Q4: What types of screws are compatible with this system?
The fixator is compatible with 6.0mm and 5.0mm screws, available in both Stainless Steel and Titanium Alloy.
Q5: Is the Multi-ring Combined Fixator provided sterile?
The standard transport package is a non-sterile paper-plastic roll bag, allowing for sterilization according to local hospital protocols.
Q6: Do you provide OEM manufacturing services?
Yes, we are a major OEM supplier for several of the largest global medical device companies, utilizing high-precision German and American machining centers.




















 Marin-one Medical
Marin-one Medical