




1 / 5










| Still deciding? Get samples of $ ! US$ 60/Piece Request Sample |
| P/N | Product Name | Material | Size |
|---|---|---|---|
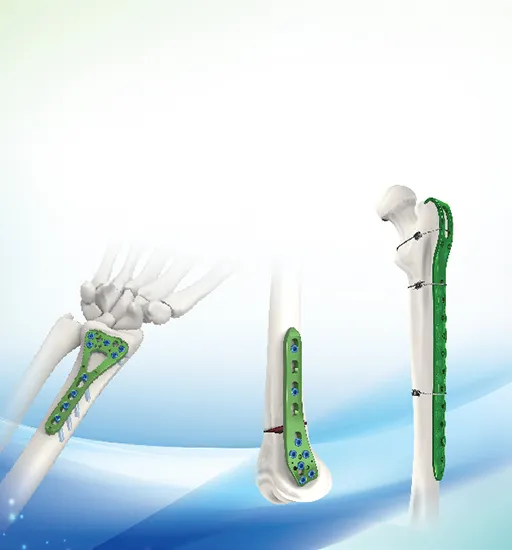
| 31102L/R | Multi-axial Distal Radius Plam Locking plate III (Left and Right types) (use 2.7 locking screws or 2.7 self-tapping screw) | Pure Titanium | 2 holes L/R |
| 31103L/R | 3 holes L/R | ||
| 31104L/R | 4 holes L/R | ||
| 31105L/R | 5 holes L/R | ||
| 31106L/R | 6 holes L/R | ||
| 31107L/R | 7 holes L/R | ||
| 31108L/R | 8 holes L/R |







 Marin-one Medical
Marin-one Medical