




1 / 5










| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
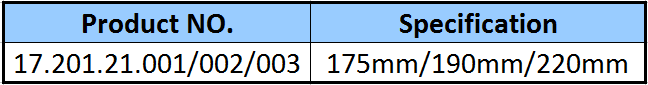
As a national high-tech enterprise, R&D investment consistently exceeds 8% of total sales. This focus on innovation has led to revolutionary technologies including:
We adhere rigorously to ISO13485 quality systems and CE MDD 93/42/EEC directives. Our production facilities feature advanced processing equipment from Germany, America, and Japan, including high-precision machining centers and automatic lathes.


Our products are exported globally to countries including Colombia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier for some of the largest global players in the medical device industry.









 Marin-one Medical
Marin-one Medical