A leading orthopedic implant manufacturer with a proven track record of innovation, quality, and global service
Guangdong Marin-one Medical Devices Co., Ltd. is specialized in manufacturing Orthopedic Implants and instruments, with a diversified model of Research and Development, production, sales, and export. The company has a perfect quality management system and advanced production capacity.
After more than 18 years of research and development, we have about 11 main product series, including the Spinal System, Intramedullary Nail System, Trauma Plate and Screw System, Locking Plate and Screw System, CMF Maxillofacial System, External Fixation, Joint System, Medical Power Tool System, General Surgical Instruments System, Sterilization Box & Basket, and Veterinary Orthopedic products.
In the principle of "Quality First, Service First, R&D First, Innovation First", the company wins an excellent reputation both domestically and abroad. Customer satisfaction is the purpose of our service.
"Since the establishment of the company, all employees have consistently adhered to the business philosophy of sincere service, continuous improvement — adhered to the service tenet of customer-centeredness, pursuit of nobility, attention to details, and reputation first. With professional after-sales service and pioneering spirit, we have established a good reputation."




Comprehensive orthopedic implant and surgical instrument solutions for hospitals, clinics, and distributors worldwide
Full range of pedicle screws, rods, cages, and accessories for cervical, thoracic, and lumbar spine surgeries.
Femoral, tibial, and humeral nails engineered for superior fixation strength and minimally invasive application.
Dynamic compression plates, reconstruction plates, and screws for complex fracture management.
Angular-stable locking plates for osteoporotic bone and periarticular fractures with predictable outcomes.
Craniomaxillofacial implants and fixation systems for facial reconstruction and cranial repair procedures.
Modular external fixators for emergency fracture stabilization, limb lengthening, and deformity correction.
Hip, knee, and shoulder joint replacement implants with high-performance bearing materials and anatomical design.
Battery-powered and pneumatic surgical drills, saws, and reamers designed for precision and reliability in the OR.
A comprehensive set of retractors, forceps, scissors, and specialty tools for general and orthopedic surgery.
Durable, autoclave-compatible sterilization containers and baskets ensuring safe instrument storage and transport.
Specialized implants and instruments for small and large animal orthopedic surgeries, trusted by veterinary professionals globally.
State-of-the-art facilities with precision CNC machining, clean-room assembly, and rigorous quality inspection




What sets Marin-one Medical apart from other orthopedic implant manufacturers
Our dedicated research and development team has over 18 years of experience in orthopedic biomechanics, material science, and implant design, continuously pushing the boundaries of surgical outcomes.
With 11 comprehensive product series covering spinal, trauma, joint, CMF, and veterinary applications, we offer a one-stop sourcing solution for hospitals and distributors, reducing supply chain complexity.
Our facility is equipped with high-precision CNC machining centers, 5-axis milling machines, and surface treatment lines, ensuring dimensional accuracy and biocompatibility of every implant produced.
We have successfully exported to over 60 countries across Asia, Europe, the Americas, the Middle East, and Africa. Our team is experienced in international regulatory requirements, documentation, and logistics.
Every product undergoes multi-stage quality inspection including dimensional measurement, mechanical testing, surface finish analysis, and biocompatibility verification before shipment.
Our dedicated after-sales team provides prompt technical support, instrument training materials, and responsive communication — ensuring our partners and end-users always receive the help they need.
Trusted by surgeons, hospitals, and distributors in over 60 countries — here is why we are the preferred partner
Our implants are developed in close collaboration with experienced orthopedic surgeons to ensure clinical relevance, ease of use, and optimal patient outcomes.
As a direct manufacturer with vertically integrated production, we offer highly competitive pricing without compromising on quality — maximizing your distribution margins.
With large in-stock inventory and efficient production scheduling, we ensure fast order fulfillment and on-time delivery to keep your supply chain running smoothly.
We offer full OEM and ODM services — from custom implant design and private labeling to custom packaging — tailored to your brand and market requirements.
Our products meet ISO 13485, CE, and CFDA standards. We assist customers with registration documentation and regulatory submissions in their target markets.
Every client receives a dedicated account manager fluent in English and experienced in international medical device trade, ensuring seamless communication and service.
Our commitment to quality is backed by internationally recognized certifications and regulatory approvals
Our quality management system is certified to ISO 13485, the international standard for medical device manufacturers, ensuring consistent product quality and safety.
Selected product series carry the CE marking under EU Medical Device Regulations, enabling direct market access across European Union member states.
All products are registered with China's National Medical Products Administration (NMPA), ensuring compliance with domestic regulatory requirements.
All implant materials undergo rigorous biocompatibility testing per ISO 10993 standards, ensuring patient safety and tissue compatibility in clinical applications.
Build your brand on our world-class manufacturing foundation — we bring your orthopedic product vision to life
Our engineering team works closely with you to understand your product specifications, target market regulations, and branding requirements.
We develop 3D CAD designs, produce rapid prototypes, and conduct biomechanical testing to validate the implant design before full production.
We prepare comprehensive technical files, test reports, and regulatory dossiers to support your product registration in target markets.
Full-scale production with 100% dimensional inspection, surface finish verification, and packaging quality control before shipment.
We offer complete private labeling services including custom logo engraving, branded packaging, and IFU document customization.
Real-world partnerships that demonstrate our technical expertise and global service capabilities
A leading German orthopedic distributor partnered with Marin-one to develop a private-label spinal fixation system. We delivered full CE-certified OEM production including custom instrument sets, branded packaging, and multi-language surgical technique guides within 8 months from design approval.
Spinal System · OEM · CE CertifiedA hospital procurement group in Brazil selected Marin-one as their primary supplier for trauma plates, screws, and intramedullary nails. We fulfilled an annual contract of over 10,000 units with consistent quality, on-time delivery, and full ANVISA documentation support.
Trauma System · Bulk Supply · ANVISAAn established UAE medical device distributor built their orthopedic portfolio around Marin-one's product range — covering spinal, trauma, joint, and external fixation systems. We provided regulatory registration support, product training, and ongoing technical assistance.
Full Portfolio · Distribution · TrainingFeedback from surgeons, distributors, and hospital procurement teams around the world
The values and principles that guide every decision we make at Marin-one Medical
We never compromise on quality. Every implant is manufactured to the highest standards to protect patient safety and surgical success.
Sincere, responsive, and professional service is the cornerstone of every long-term relationship we build with our global partners.
Continuous investment in research and development keeps our product portfolio at the forefront of orthopedic innovation and clinical need.
We embrace new technologies, materials, and manufacturing processes to deliver better implants and instruments for the surgeons of tomorrow.
Whether you are a distributor, hospital procurement team, or looking for a reliable OEM manufacturer — we are here to help you succeed. Contact our team today for a product catalog, pricing, or custom OEM consultation.
📩 Contact Us Today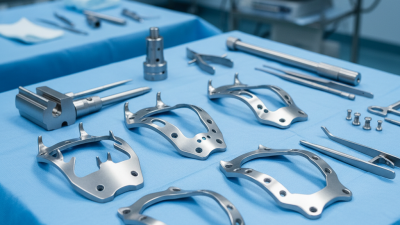
In the evolving world of orthopedic implants, Clavicle Hook Plates have emerged as essential tools for surgeons. They provide support and stability
Author: Liam Date: 2026-03-30
Learn More
At the 2026 Canton Fair, global buyers will discover the finest "Orthopedic L Plates" among various innovative products. This major event is
Author: Ethan Date: 2026-03-27
Learn More
The upcoming Canton Fair in 2026 presents a unique opportunity for professionals in the medical device industry, particularly those focused on
Author: Sophia Date: 2026-03-25
Learn More